Dabertrand Lab

Primary Appointment: Associate Professor, Anesthesiology
Secondary Appointment: Associate Professor, Pharmacology
Education
- BS Structural Biochemistry, University of Bordeaux Segalen, France
- BS Cell Biology and Physiology, University of Bordeaux Segalen, France
- MS Neuroscience and Neuropharmacology, University of Bordeaux Segalen, France
- Ph.D. Neuroscience and Neuropharmacology, University of Bordeaux Segalen, France
- Postdoctoral Fellow, Center for Cognitive and Integrative Neurosciences, University of Bordeaux Segalen, France
- Postdoctoral Fellow, Pharmacology, Larner College of Medicine University of Vermont, VT
Contact Information
Mail Stop B112
12700 E. 19th Avenue, room P15-7590A
Aurora, Colorado 80045
Telephone: (303) 724-3792
Email: [email protected]
Website
Training Programs
Medical Scientist Training
Neuroscience
Pharmacology
Research Interests
The control of cerebral blood flow by ion channels and calcium signaling in the pericytes, endothelial cells, and smooth muscle cells that constitute the parenchymal microcirculation, and use this information to combat brain diseases with a vascular component.
Research Description
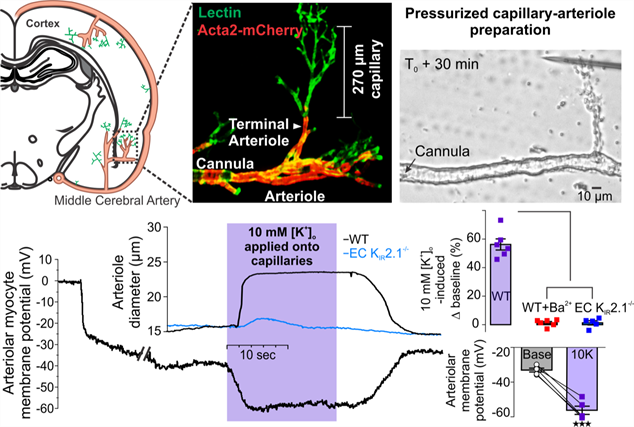
The overall goal of our research is to understand the control of cerebral blood flow in physiological and small vessels disease conditions, with a special interest in the interaction between neural activity and the microvasculature. Our early studies focused on the control of smooth muscle and endothelial cell function by ion channels, providing some of the first measurements of membrane potential, calcium signaling, and diameter imaging of mouse pressurized parenchymal arterioles. Developing these approaches in murine microcirculation was a tour de force that lets us to use genetic mouse models and investigate potential treatments for cerebrovascular diseases. These accomplishments lead to several article highlights and awards including the New Investigator Award from The American Physiological Society in 2015. The emerging view from our work on CADASIL, an archetypal monogenic form of small vessel disease of the brain, is that fundamental alterations in extracellular matrix proteins disrupt the ion channel repertoire controlling vascular reactivity, and then cerebral hemodynamics, at a very early stage. In our more recent work on neurovascular coupling, the mechanism sustaining functional hyperemia, we established brain capillaries as an active sensory web detecting neural activity and communicating it to upstream arterioles in the form of vasodilatory signals to match the local metabolic demands. Currently, our federally funded work (NHLBI/NIH) is aimed at understanding the disruption of neurovascular coupling in disorders involving extracellular matrix alterations to shed light on vascular dementia.
Publications
Click here for Dr. Fabrice Dabertrand's publications on NCBI