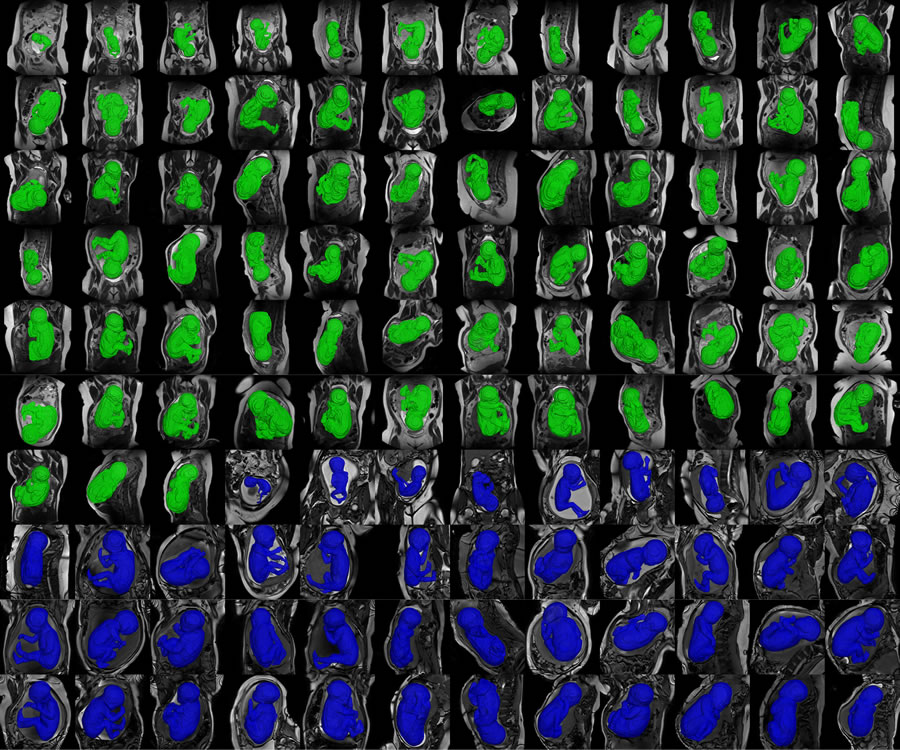
Radiology Research
The Department of Radiology supports both clinical and basic science research and leads the Colorado University Research Imaging Center (CURIC). The Quantitative Image Analysis (QIA) Laboratory (formerly BIRL) provides design and analysis services for imaging studies. Clinical research performed at the University of Colorado and requiring imaging is approved and administered through the Department of Radiology.