Aaron Johnson, PhD
Associate Professor

Education:
Ph.D., Rockefeller University
Contact Information:
Phone: (303) 724-3224
E-mail: [email protected]
Graduate Program Memberships:
Research in the lab
Our work focuses on the formation and regulation of chromatin domains and their ultimate roles in epigenetic genome regulation. We are particularly interested in the mechanisms of heterochromatin establishment and function. Heterochromatin operates in organisms from yeast to humans to determine cell identity and maintain genome stability by silencing genes. Because heterochromatin functions in such central processes, misregulation of this genomic structure can have dire consequences such as cancer or abnormal development. Our work investigates the mechanisms by which silencing is carried out. We focus on two pathways: long noncoding RNA- and sirtuin-mediated heterochromatin. Our group approaches the study of these pathways by a combination of in vitro assembly of chromatin domains, mechanistic biochemistry, proteomic analysis, and genome-wide chromatin profiling to understand the complex superstructural “neighborhoods” of chromosomes.
Our group focuses on two main projects:
1) Uncovering mechanisms of long noncoding RNA(LncRNA)-mediated gene silencing
It has recently been appreciated that the majority of the human genome has the capacity to be transcribed into RNA. Some of these non-protein coding RNA products, made from what was once considered "junk DNA," can have specific functions in
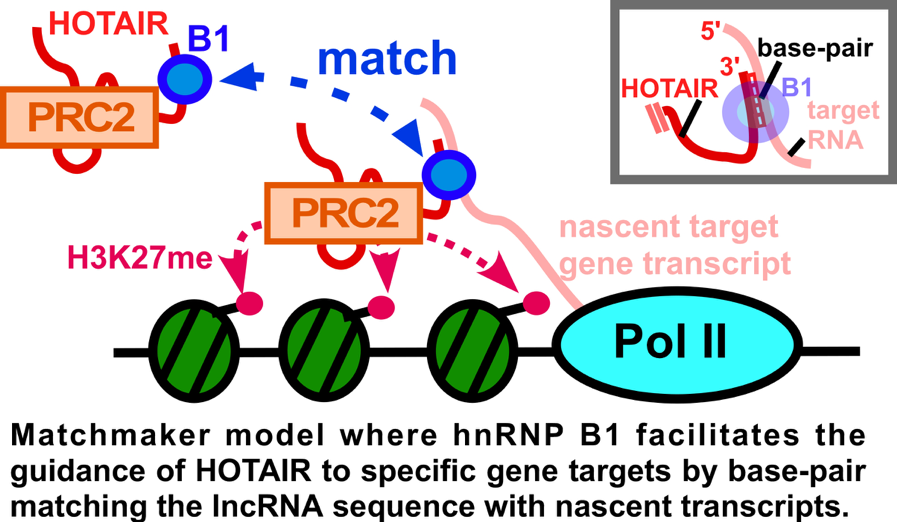
the cell, such as contributing to gene regulation. We have focused on investigating the molecular mechanism behind long noncoding RNAs that regulate heterochromatin. Recently, we have generated a new model for how a LncRNA called
HOTAIR may choose its target genes via RNA-RNA matchmaking. HOTAIR has roles in developmental patterning and overexpression in cancer leads to aberrant gene silencing that promotes aggressive cancer characteristics. By studying
how LncRNAs work at the molecular level, we have a shot at figuring out how to control them in disease to reset normal epigenetic states.
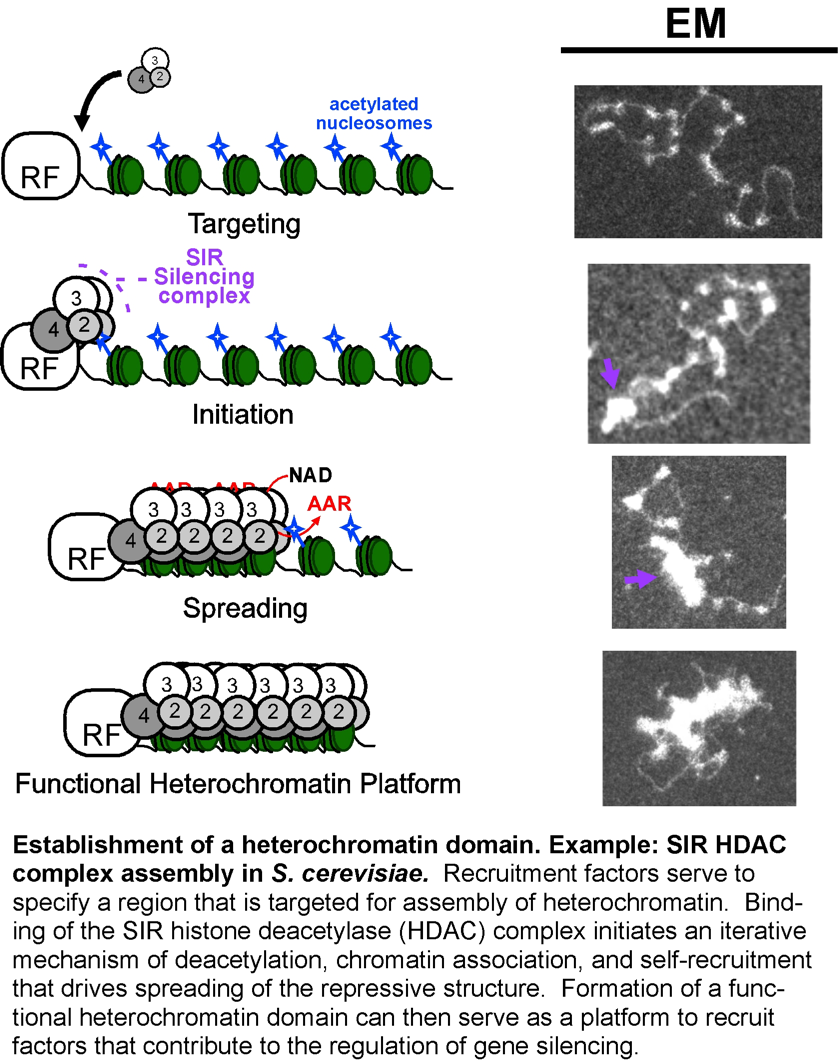
2) Using budding yeast heterochromatin as a model to comprehensively characterize a specific chromatin domain.
The genetics of budding yeast chromatin-mediated gene silencing have provided an extensive foundation for understanding chromatin biology in all eukaryotes. This model system is distinguished by the minimal set of components necessary for gene silencing. The next step for this model is to provide new insights through biochemistry. We have demonstrated that a functional yeast heterochromatin domain can be assembled in the test tube, and uncovered how versatile heterochromatic gene silencing is, even in its most stripped-down form in budding yeast.
Projects include:
- The interplay of heterochromatin and DNA replication
- Detailed biochemical analysis of interactions between RNA Polymerase II and silencing factors.
- Understanding the mechanism of directional spreading of the silencing structure along a chromatin fiber.
- How heterochromatin is re-established in an epigenetic pattern.
- Comprehensive characterization of the yeast heterochromatin interactome using quantitative proteomics.

| Pictures | First Name | Last Name | Job Title | |
|---|---|---|---|---|
 | Jenna | Kotz | Graduate Student | [email protected] |
 | Edgardo | Linares | Graduate Student | [email protected] |
 | Francisco | Perez | Postdoctoral Scholar | [email protected] |
 | Sierra | Simmerman | Graduate Student | [email protected] |