Tumor Cell Plasticity and Metastasis
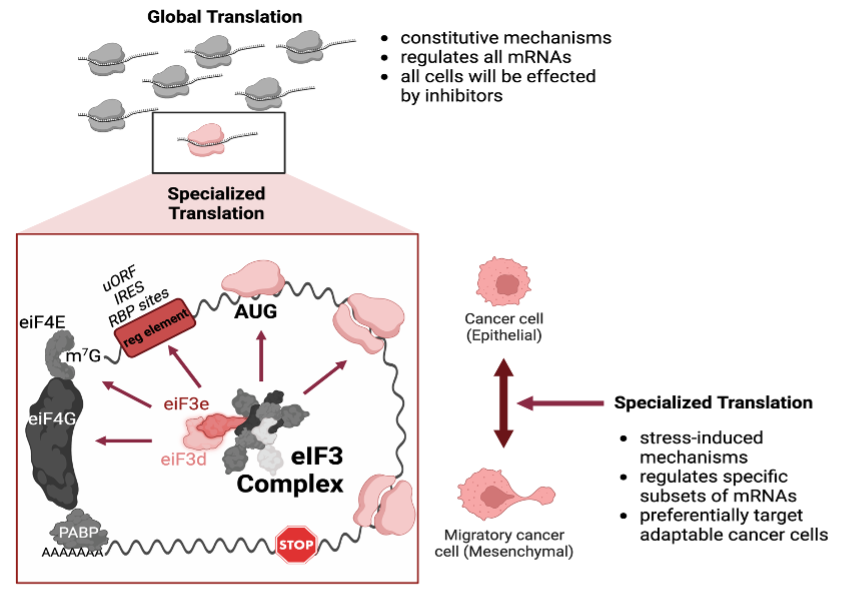
Virtually all breast cancer associated deaths occur due to metastasis. During this multi-step process, the surviving cancer cells must rapidly adapt to many stressors, including hypoxia, lack of cell-cell/matrix interactions, inflammation, immune surveillance, and chemotherapy. Many studies have shown that genetic alterations do not play a dominant role in metastasis. Instead, epigenetic/transcriptional changes have emerged as key means by which tumor cells rapidly respond to stressors during the metastatic cascade. Recently, several groups (including the University of Colorado Anschutz Cancer Center) have shown that in addition to transcription, stress-induced mRNA translation may play a much larger role in tumor cell adaptation than was previously appreciated.
With this scientific focus group, our goal is to test the overarching hypothesis that translational changes in response to microenvironmental stressors drives cancer cell adaptation and survival, resulting in metastasis. Identifying both unique and shared mechanisms of tumor cell adaptation in response to different stressors may enable us to uncover specific means to target breast cancer metastasis. To address this hypothesis, we are working on four different projects:
Project 1
To determine whether translational regulation by eIF3d and eIF3e mediates the hypoxic response, breast cancer cell adaptability and metastasis.
Project 2
To understand how translation regulates adaptation to radiotherapy-induced stress to promote radioresistance in brain metastasis.
Project 3
To determine how pulmonary viral infections alter translational regulation in the lung to promote metastatic outgrowth.
Project 4
To determine how translation in response to chemotherapy induced ER stress contributes to tumor progression and metastasis.
These four projects will be supported by the Multi-Omics Data Integration and Modeling Core. The core will support experimental design for -omic data generation, develop robust and reproducible analysis pipelines, develop methods to integrate multi-omics datasets, and generate novel molecular signatures to evaluate translational changes across projects.
Project Leads

Heide Ford, PhD
Tumor Cell Plasticity and Metastasis