Shared Content Block:
Styles for CDB Faculty Pages
Tom Finger, Ph.D.
Professor

| [email protected] | |
| 303-724-3436 | |
| Ph.D. — Massachusetts Institute of Technology, 1975 | |
| Rocky Mountain Taste and Smell Center |
Development and Organization of the Chemical Senses
The chemical senses, including taste, smell and trigeminal sensitivity, play an important role in social and ingestive activities of all animals. A major emphasis in my laboratory is on questions concerning the development and organization of these systems in several vertebrate models. Three major areas are under investigation at present: 1) morphology and function of solitary chemosensory cells (see Finger et al. PNAS 2003) in protection of the airways and gut, 2) the cellular organization and development of taste buds (see Finger et al, Science 2005, and 3) regulation of feeding behavior by taste and other oropharyngeal chemoreceptors.
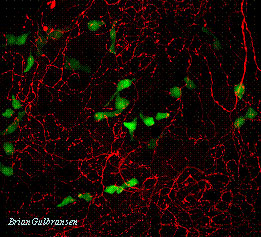
Nasal solitary chemosensory cells (green) innervated by trigeminal nerve fibers (red).
Solitary Chemosensory Receptor Cells are situated in the respiratory epithelium of the nasal cavity and make contact with sensory fibers of the trigeminal nerve. These chemoreceptor cells utilize diverse receptor mechanisms to detect noxious substances in the incoming airstream and elicit protective reflexes such as sneezing, coughing or cessation of inspiration (apnea). We are investigating how these cells differentiate from epithelial basal cells and how they detect the presence of noxious substances.
Taste buds consist of short, neuroepithelial receptor cells embedded in gustatory epithelia, e.g. of the tongue, palate and larynx. We have shown that taste buds consist of 3 distinct differentiated cell types each serving a distinct function (Yee et al., 2001; Bartel et al, 2006). Our lineage analysis in transgenic mice, shows that taste buds arise from local epithelium and originate from a handful of epithelial progenitors (Stone et al. 1995). Our current experiments examine the role of each type of cell in detection of different taste qualities.
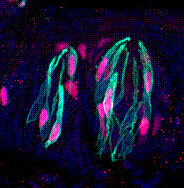
Taste buds comprise 3 distinct cell types, each with a different function.
Feeding behavior is regulated largely by gustatory cues in many vertebrates. In order for a potential foodstuff to be swallowed, it must trigger an appetitive gustatory cue. Taste buds release ATP to activate the gustatory nerves and genetic elimination of ATP receptors (P2X2 and P2X3) on the gustatory nerves results in a mouse unable to taste anything (Finger et al 2005). Yet these animals maintain normal weight and can still react when they ingest certain substances. We are investigating the role of taste and non-taste chemoreceptors in the oropharynx and gut in regulation of food intake in these “tasteless” mice (Hallock et al, 2009).
We also use non-mammalian models to study central processing and transmission of taste information. Goldfish have evolved an elaborate specialization of the pharynx that is involved in sorting food from substrate (Lamb & Finger 1995; Sharp & Finger, 2002). The neuronal machinery involved in this behavior is situated in an easily accessible, laminated structure of the hindbrain. This organization is conducive to in vitro slice physiology and pharmacology as well as to in Ca++ imaging studies on synaptic transmission (Ikenaga et al. 2009).
View Dr. Finger's Latest Publications in PubMed
Stone LM, Finger TE, Tam PP, Tan SS. 1995. Taste receptor cells arise from local epithelium, not neurogenic ectoderm. Proc Natl Acad Sci U S A 92:1916-1920.
Finger TE, Böttger B, Hansen A, Anderson KT, Alimohammadi H, Silver WL. 2003. Solitary chemoreceptor cells in the nasal cavity serve as sentinels of respiration. Proc Natl Acad Sci U S A 100:8981-8986.
Hall JM, Bell ML, Finger TE. 2003. Disruption of sonic hedgehog signaling alters growth and patterning of lingual taste papillae. Dev Biol 255:263-277.
Yee CL, Jones KR, Finger TE. 2003. Brain-derived neurotrophic factor is present in adult mouse taste cells with synapses. J Comp Neurol 459:15-24.
Finger TE, Danilova V, Barrows J, Bartel DL, Vigers AJ, Stone L, Hellekant G, Kinnamon SC. (2005) ATP signaling is crucial for communication from taste buds to gustatory nerves. Science 310:1495-9.
Gulbransen BD, Finger TE. 2005. Solitary chemoreceptor cell proliferation in adult nasal epithelium. J Neurocytol 34:117-122.
Finger TE (2007) Sorting Food from Stones: The Vagal Taste System in Goldfish, Carassius auratus. J . Comp. Physiol. A. 194(2):135-143. PubMed / Full Text
Tizzano M, Dvoryanchikov G, Barrows JK, Kim S, Chaudhari N, Finger TE. 2008. Expression of Galpha14 in sweet-transducing taste cells of the posterior tongue. BMC Neurosci. 2008 Nov 13;9:110. PubMed / Full Text
Finger, T.E. (2009) Evolution of Gustatory Reflex Systems in the Brainstems of Fishes. Integr Zool 4: 53-63. Full Text
Hallock, RM, Martyniuk, CJ ,and Finger TE (2009) Group III Metabotropic Glutamate Receptors (mGluRs) Modulate Transmission of Gustatory Inputs in the Brainstem. J Neurophysiol. 102(1):192-202. PMID: 19369363 PubMed / Full Text
Ikenaga, T., Ogura, T., and Finger , TE (2009) Vagal gustatory reflex circuits for intraoral food sorting behavior in the goldfish; Cellular organization and neurotransmitters. J. Comp. Neurol. 516(3):213-25. PubMed / Full Text.
Hallock RM, Tatangelo M, Barrows J, & Finger TE (2009) Residual chemosensory capabilities in double P2X2/P2X3 purinergic receptor null mice: intraoral or postingestive detection? Chem Senses 34(9):799-808. Pub Med
Tizzano M, Gulbransen BD, Vandenbeuch A, Clapp TR, Herman JP, Sibhatu HM, Churchill ME, Silver WL, Kinnamon SC, Finger TE. 2010. Nasal chemosensory cells use bitter taste signaling to detect irritants and bacterial signals. Proc Natl Acad Sci U S A 107:3210-3215.