Shared Content Block:
Styles for CDB Faculty Pages
Maureen Stabio, Ph.D.
Associate Professor
Vice Director, Modern Human Anatomy Program

| [email protected] | |
| 303-724-7461 | |
| Ph.D., Anatomy and Neurobiology, Boston University School of Medicine, 2007 | |
| CU Plastinated Organ Library |
Graduate Program Affiliations:
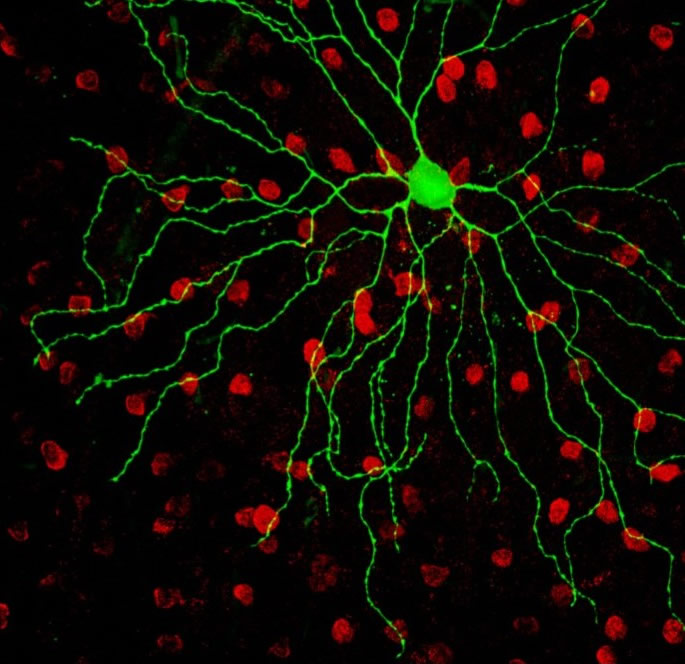
An M4 subtype of intrinsically photosensitive retinal ganglion cell (green) sits in a sea of starbust amacrine cells (red) in the mouse retina. The M4 cell was dialyzed with lucifer yellow during patch clamp recording and the starburst amacrine cells were immunostained for Choline Acetyltransferase. Scale bar = 50 microns
My research interest and expertise is in the structure and function of the retina. My particular focus is on retinal ganglion cells (RGCs), which serve as conduits of light information from the retina to the brain. There are up to 20 different types of RGCs that carry different types of information, such as color, form, motion or contrast, and project to different brain regions to drive different behaviors. One of the major goals of visual neuroscience is to define the diversity of RGC subtypes and to map their complex circuitry. One recently-discovered RGC subtype is unique from all others in that it contains its own photopigment (melanopsin) and can send light signals to the brain independent of rod and cones.
These intrinsically photosensitive retinal ganglion cells (or ipRGCs) are important for subconscious or reflexive visual behaviors such as circadian photoentrainment and the pupillary light reflex. IpRGCs have fascinated the vision science community since their discovery a little over a decade ago, and five morphologically and physiologically distinct subtypes of ipRGCs (named in order of their discovery as M1-M5) have been discovered since. During my post-doctoral training in the Berson Lab at Brown University, I characterized the form and function of the M4 and M5 subtypes of ipRGCs in the mouse retina and discovered that, unlike previously characterized ipRGCs, M4 and M5 cells have features implicating them in mechanisms of pattern vision.
My current research translates my expertise in retinal structure and function to study retinal disease. Interestingly, different retinal cell types die in specific sequences in many eye diseases. For example, in some diseases, ipRGCs have been found to be resistant to cell death while other RGCs progressively degenerate. Understanding the diversity of RGC types is important for developing therapies for vision restoration that involve genetic modification or electrical stimulation of RGCs. I am currently working in collaboration with the Lefcort lab at Montana State University to investigate the RGC types (and other retinal cell types) that degenerate in mouse models of Familial Dysautonomia (FD). FD is a devastating disease of the sensory and autonomic nervous system that causes multiple neuropathies in patients; one of the most debilitating is progressive visual loss that leads to complete blindness. Our long-term goal is to cure and prevent blindness in FD patients through a gene therapy approach. The first step towards this goal (experiments which we are currently conducting in my laboratory) is to determine the retinal cell types that degenerate in mouse models of FD.
Educational Scholarship
In addition to my research in the laboratory, I am also an experienced teacher and have been trained in pedagogy and educational research through teaching certificate programs at Boston University School of Medicine and Brown University. I am most interested in designing 3D computer graphic models based on magnetic resonance imaging datasets of the human brain. My goal is to students visualize and understand 3D relationships of internal brain structures based on clinical images that are shown in 2D slices. A pilot demo I developed with a talented, former medical student mentees, Zachary Drapkin, is illustrated here: https://www.youtube.com/watch?v=5KkK9TFc3Xs. I conduct research on the effectiveness of these dynamic teaching tools compared to other static computer-based educational tools. My goal is to help students overcome their “neurophobia” (or fear of neuroanatomy) and to be able to solve clinical problems by having a thorough understanding of the 3D spatial relationships and pathways of the brain, brainstem and spinal cord.

Educational Research Publications
Feiler J, Stabio ME. 2018. Three pillars of educational neuroscience from three decades of literature. Trends in Neuroscience and Education Nov 15; 13; 17-25.
R. Klaus, D. Royer, M.E. Stabio . Use and perceptions of plastination among medical anatomy educators in the United States. Clinical Anatomy.Mar;31(2):282-292. doi: 10.1002/ca.23025. Epub 2017 Dec 27.
R.J. Hlavac, K. Betts, R.Klaus, M.E. Stabio. Novel dissection of the central nervous system to bridge gross anatomy and neuroscience for an integrated medical curriculum. Anatomical Sciences Education 2017 Aug 17. doi: 10.1002/ase.1721. [Epub ahead of print]
Z.A. Drapkin, K.A. Lindgren, M. Lopez, M.E. Stabio. Development and assessment of a new 3D neuroanatomy teaching tool for MRI training. Anatomical Sciences Education. 2015; 8 (6): 502-9
M.E. Estevez, K.A. Lindgren, P.R. Bergethon. A novel 3-dimensional tool for teaching human neuroanatomy. Anatomical Sciences Education. 2010, 3: 309-317.
M.E. Estevez. Mentorship in Graduate Education. Change Jan/Feb 2009, 55-56.
Scientific Research Publications
Quattrochi LE, Stabio ME, Kim I, Ilardi MC, Fogerson PM, Leyrer M, Berson DB. 2018. The M6 cell: a small-field bistratified photosensitive ganglion cell. J Comp Neuro 2019 Jan 1;527(1):297-311. doi: 10.1002/cne.24556.
Sondereker KB, Stabio ME, Renna JM. Crosstalk: The diversity of melanopsin ganglion cell subtypes challenges the canonical divide between subconscious and conscious vision. J Comp Neuro. 2020 Aug;528(12):2044-2067. doi: 10.1002/cne.24873. Epub 2020 Feb 18.
Sondereker KB, Stabio ME, Jamil JR, Tarchick MJ, Renna JM. 2018. Where You Cut Matters: A Dissection and Analysis Guide for the Spatial Orientation of the Mouse Retina from Ocular Landmarks. J Vis Exp. Aug 4;(138). doi: 10.3791/57861.
Stabio ME, Sondereker KB, Haghgou SD, Day BL, Chidsey B, Sabbah S, Renna JM. 2018. A novel map of the mouse eye for orienting retinal topography in anatomical space. J Comp Neuro. 2018 Apr 6. doi: 10.1002/cne.24446. [Epub ahead of print].
M.E. Stabio, L. Quattrochi, M.C. Ilardi, P.M. Fogerson, J.R. Renna, M.T. Kim, I. Kim, M. Schiel, K.L. Briggman, D.M. Berson. The M5 Cell: A color-opponent intrinsically photosensitive retinal ganglion cell. Neuron. Jan 3; 97(1):150-163.e4.
P.J. Bonezzi, M.E. Stabio, and J.M. Renna. The development of medium wavelength photoresponsivity in the postnatal mouse retina. Current Eye Research 43 (5); 666-673. doi: 10.1080/02713683.2018.1433859.
Y. Ueki, G. Ramirez, E. Salcedo, M.E. Stabio, F. Lefcort. Loss of Ikbkap causes slow, progressive retinal degeneration in a mouse model of Familial Dysautonomia. eNeuro. 2016. 3(5) e0143-16.
J.R. Renna, D.K. Challappa, C.L. Ross, M.E. Stabio, D.B. Berson, Melanopsin ganglion cells extend dendrites into the outer retina during early postnatal development, Developmental Neurobiology, 2015 Sep;75(9):935-46.
J.-S. Wang, S. Nymark, R. Frederiksen, M. Estevez, S. Shen, J. Corbo, M.C. Cornwall, V. Kefalov, Chromophore supply rate-limits mammalian photoreceptor dark adaptation. 2014 J. Neuroscience. 2014, 34(34): 11212-11221.
O. Dhande, M.E. Estevez†, L.E. Quattrochi, R. El-Danaf, P. Nguyen, D.M. Berson, A. Huberman. Genetic dissection of retinal inputs to brainstem nuclei controlling image stabilization. J Neuroscience. 2013, 33(45):17797-17813.
S. Weng, M.E. Estevez, D.M. Berson, Mouse ganglion-cell photoreceptors are driven by the most sensitive rod pathway and by both types of cones. PLoS ONE, 2013, 8(6): e66480.
M.E. Estevez, P.M Fogerson, M.C. Ilardi, B.G. Borghuis, E. Chan, S. Weng, O.N. Auferkorte, J.B. Demb, D.M. Berson. Form and Function of the M4 Cell, an Intrinsically Photosensitive Retinal Ganglion Cell Type Contributing to Geniculocortical Vision. J Neuroscience. 2012, 32(39):13608-20.
M.E. Estevez, A.V. Kolesnikov, P. Ala-Laurila, R.K. Crouch, V.I. Govardovskii, and M.C. Cornwall. The 9-methyl group of retinal is essential for rapid Meta II decay and phototransduction cascade quenching in red cone photoreceptors. 2009, J Gen Physiol. 134:137-150.
J.-S Wang, M.E. Estevez, M.C. Cornwall, V.J. Kefalov. Intra-retinal visual cycle required for rapid and complete cone dark adaptation. Nat. Neuroscience. 2009, 12:295-302.
A.V. Kolesnikov, P. Ala-Laurila, S.A. Shukolyukov, R.K. Crouch, B. Wiggert, M.E. Estevez, V.I. Govardovskii, M.C. Cornwall. Visual cycle and its metabolic support in gecko photoreceptors. Vision Res. 2007. 47:363-374
M.E. Estevez, P. Ala-Laurila, R.K. Crouch, M.C. Cornwall. Turning cones off: the role of the 9-methyl group of retinal in red cones. J Gen Physiol. 2006, 128:671-685.
Q. He, D. Alexeev, M.E. Estevez, S.L. McCabe, P.D. Calvert, D.E. Ong, M.C. Cornwall, A.L. Zimmerman, C.L. Makino. Cyclic nucleotide-gated ion channels in rod photoreceptors are protected from retinoid inhibition. J Gen Physiol. 2006, 128:473-485.
P. Ala-Laurila, A.V. Kolesnikov, R.K. Crouch, E. Tsina, S.A. Shukolyukov, V.I. Govardovskii, Y. Koutalos, B. Wiggert, M.E. Estevez, M.C. Cornwall. Visual cycle: dependence of retinol production and removal on photoproduct decay and cell morphology. J Gen Physiol. 2006, 128:153-169.
V.J. Kefalov, M.E. Estevez, P. Goletz, M. Kono, R.K. Crouch, M.C. Cornwall, K.-W. Yau. Breaking the covalent bond -- a pigment property that contributes to desensitization in cones. Neuron 2005, 46, 879-890.
“It's Not Your Fault”: CU Anschutz Students Talk With Teens About Mental Health (CU Anschutz Today, February 2020)
Little Understood Cell Helps Mice See Color (Featured on NeuroscienceNews.com, Science Daily, University of Akron Online, and CU Anschutz Today, Dec 2017)
Guide for en bloc cadaveric extraction of the central nervous system (Anatomy Now: Official Newsletter of the American Association of Anatomists, September 2017)
Remarkable en bloc dissection of human central and peripheral nervous system accomplished at University of Colorado (Neuropathology Blog, June 2017)
Middle schoolers dissect brains, consider health care careers (CU Anschutz Today, February 2017)