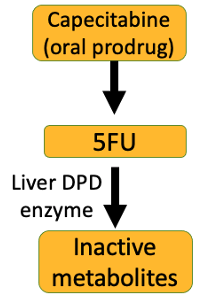
Fluoropyrimidines, including 5-fluorouracil (5-FU) and capecitabine, are agents used in the treatment of solid tumors, such as gastrointestinal, breast, and head and neck cancers. DPD, an enzyme in the liver, metabolizes most of 5-FU to inactive metabolites.
DPYD is a gene that encodes the DPD liver enzyme. Variation in the DPYD gene results in three DPD metabolizer phenotypes: normal, intermediate, and poor metabolizers.
The DPD intermediate metabolizer phenotype is observed in approximately 2-8% of the population. The DPD poor metabolizer phenotype is rare, observed in less than 0.2% of the population. Intermediate and poor metabolizer phenotypes are associated with decreased DPD activity and an increased risk of severe or even fatal drug toxicity with systemic fluoropyrimidine drugs.
Most research has focused on systemic fluoropyrimidines and DPYD genotypes. However, 5-FU is also used as a topical formulation, primarily for the treatment of actinic or solar keratoses and superficial basal cell carcinoma. A case report suggests that patients with the poor metabolizer phenotype may have an increased risk of severe toxicity when prescribed topical fluorouracil (5-FU).