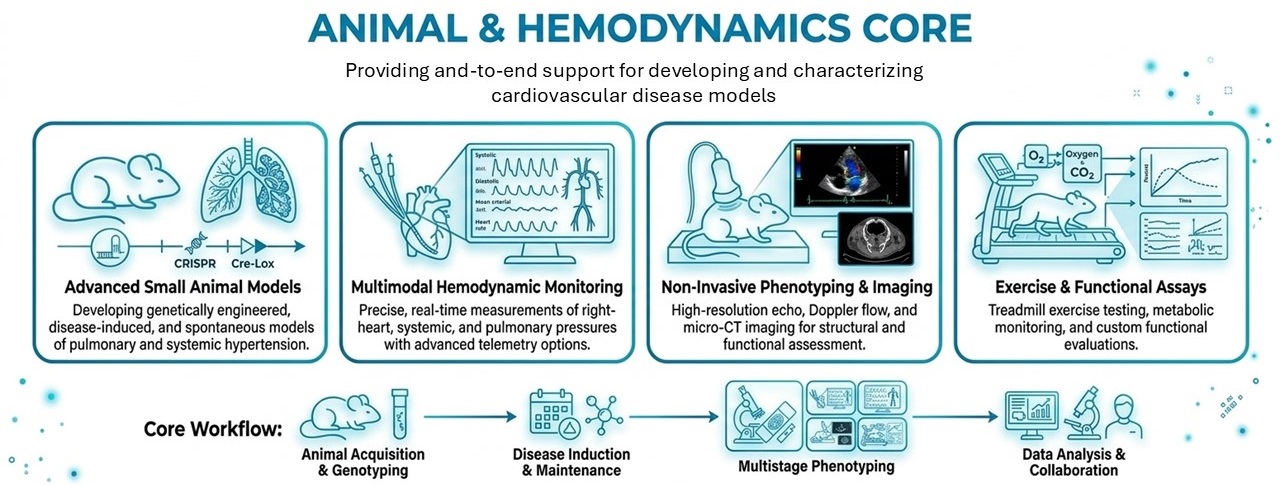
The Animal and Hemodynamics Core provides a comprehensive platform for the development, maintenance, and physiological characterization of experimental models of pulmonary vascular disease. This core supports investigators across the CVP by enabling rigorous, standardized assessment of disease progression, cardiovascular function, and therapeutic response in both small and large animal models.
By integrating genetic engineering, animal physiology, and advanced hemodynamic measurements, the core allows investigators to define how molecular and cellular mechanisms translate into functional changes in pulmonary vascular disease. The core operates in close coordination with the Histopathology and Spatial Transcriptomics Cores, ensuring that physiological phenotypes are directly linked to structural and molecular changes in the pulmonary vasculature.
Core Mission
The mission of the Animal and Hemodynamics Core is to provide investigators with:
- Robust and reproducible animal models of pulmonary hypertension (PH)
- Standardized hemodynamic and physiological phenotyping
- Quantitative assessment of exercise capacity and cardiovascular function
- Integrated workflows connecting animal models to tissue pathology and molecular profiling
- Expert support in experimental design, animal protocols, and data interpretation
This integrated approach ensures that experimental findings are physiologically meaningful and translationally relevant, supporting studies across multiple PH etiologies and disease models.
Core Capabilities
Animal Model Development and Husbandry
The core provides full support for the development and maintenance of experimental models of pulmonary hypertension, including:
- Generation of genetically engineered murine models (e.g., conditional knockouts and overexpression systems)
- Breeding, genotyping, and colony management of transgenic mouse lines
- Support for multiple PH models, including:
- Chronic hypoxia models
- SU5416 + hypoxia models
- Monocrotaline models
- Sickle cell disease-associated PH models
- Schistosomiasis-associated PH models
- Large animal (calf) hypoxia models
These models allow investigators to study disease mechanisms across a spectrum of clinically relevant conditions.
Hemodynamic and Cardiovascular Phenotyping
A central strength of the core is its ability to provide high-resolution, quantitative assessment of cardiovascular function across species.
Capabilities include:
- Measurement of right ventricular and pulmonary artery pressures
- Pressure–volume loop analysis using solid-state catheter systems (Millar, Scisense)
- Cardiac output assessment using dye dilution techniques
- Blood gas analysis and systemic hemodynamic monitoring
- Continuous and longitudinal hemodynamic measurements using telemetry systems
These approaches provide precise evaluation of disease severity, progression, and response to interventions.
Exercise Physiology and Functional Assessment
The core provides quantitative measures of functional capacity in animal models, including:
- Exercise tolerance testing using treadmill-based systems
- Determination of critical speed, a robust metric analogous to the human 6-minute walk test
- Longitudinal assessment of functional decline or therapeutic response
Hypoxia Exposure Systems
The core maintains specialized facilities for controlled hypoxia exposure in both small and large animals:
- Hypobaric hypoxia chambers for rodent models
- Large-animal hypoxic chamber systems for calf studies
- Controlled environmental conditions for long-term disease induction
Tissue Collection and Integration with Downstream Analysis
The core provides standardized protocols for:
- Tissue and organ collection
- Blood and plasma sampling
- Lung inflation and fixation for optimal morphology
- Preparation of samples for histological, molecular, and spatial analyses
All tissue collection is performed in close coordination with the Histopathology and Spatial Transcriptomics Cores, ensuring seamless integration across platforms and maximizing the scientific value of each experiment.
Technologies and Instrumentation
The Animal and Hemodynamics Core is equipped with state-of-the-art instrumentation, including:
- Solid-state catheter systems (Millar, Scisense)
- Echocardiography platforms for non-invasive cardiac assessment
- Hemodynamic data acquisition systems (BioPac, telemetry systems)
- Blood gas analyzers
- Small animal treadmill systems
- Dye dilution systems for cardiac output measurement
- Hypobaric and hyperbaric chamber systems
Expertise and Leadership
The core is led by a highly experienced team with extensive expertise in pulmonary vascular biology, animal physiology, and translational research.
- Dr. David Irwin (Director) – expert in hypoxia physiology and pulmonary hypertension models
- Dr. Cheng Jun Hu (Co-Director) – specialist in genetic engineering and transgenic mouse models
- Dr. Greta Krafsur – veterinary pathologist with expertise in large-animal models and investigative pathology
The core team includes highly skilled technical staff with expertise in:
- Animal surgery and catheterization
- Hemodynamic data acquisition and analysis
- Exercise physiology testing
- Animal handling and colony management
- Tissue and organ collection
Supporting Translational Research
The Animal and Hemodynamics Core plays a central role in linking molecular mechanisms to physiological outcomes in pulmonary vascular disease.
By integrating with the CVP’s Histopathology and Spatial Transcriptomics Cores, the Animal Core enables investigators to:
- Connect molecular pathways with functional disease phenotypes
- Evaluate therapeutic interventions in preclinical models
- Translate findings from animal models to human pulmonary hypertension
This coordinated approach ensures that discoveries are not only mechanistically informative but also clinically relevant and translatable.