The Fragile X Gene
Fragile X syndrome and associated disorders are caused by a change (also called a mutation) in a gene on the X chromosome, called the FMR-1 gene. Chromosomes are made up of thousands of genes that are passed from generation to generation. Most individuals have 46 chromosomes (23 pairs), two of which are sex chromosomes. In males, the sex chromosomes are X and Y. In females there are two X chromosomes. Each gene of a chromosome contains a specific code of DNA that determines the function of the gene in the body and produces a protein. The FMR-1 gene produces a protein called FMRP, which plays an important role in brain development and in the biological mechanisms involved in learning and memory. While all people have the FMR-1 gene, people who have fragile x syndrome or associated disorders carry a mutation impacting the body’s process of making the FMRP protein.
The FMR1 gene has a CGG “triple repeat” within the DNA that can expand and result in the mutation causing fragile x syndrome or associated disorders. Typically, individuals have <45 CGG repeats. A diagnosis of Fragile X syndrome is confirmed when there are >200 CGG repeats (“full mutation”), as this large expansion turns off the gene so no FMRP protein is produced. A diagnosis of a premutation carrier is confirmed when there are between 55-200 repeats (“premutation”), in which the gene produces protein, however the precursors to the protein, called mRNA, are produced in excess. The excess of mRNA can cause medical and psychological symptoms in premutation carriers. Premutation carriers may transmit either the full mutation or the premutation to their children. A diagnosis of intermediate/grey-zone occurs when there is 45-55 CGG repeats and the gene is less stable. Due to males having one X and one Y chromosome, and females having two X-chromosomes, females tend to be less affected with Fragile X due to the presence of the second X-chromosome, which is often able to continue to produce some FMRP protein.
Inheritance
There are two chromosomes (X and Y) that, in combination, determine the sex of an individual. Women have two X chromosomes, whereas men have one X chromosome and one Y chromosome. During reproduction, a child receives one sex chromosome from each parent. A mother contributes a copy of one of her X chromosomes. A father contributes a copy of either his X chromosome or his Y chromosome. If the father provides his X chromosome, the child will be female. If the father provides his Y chromosome, the child will be male. Because the FMR1 gene is located on the X chromosome, the risk of inheritance differs depending on the sex of the affected parent and the sex of the child.
The following diagram depicts the genetic inheritance of the FMR1 gene. The left side of the diagram represents the transmission of a fragile x premutation from a carrier father to his children. A father passes a copy of his Y chromosome to his sons. Because the fragile X gene is located on the X chromosome, fragile X is never passed from father to son. However, a father passes his only X chromosome to all of his daughters, and therefore all daughters inherit his premutation. The number of CGG repeats is typically stable when passed from father to daughter.
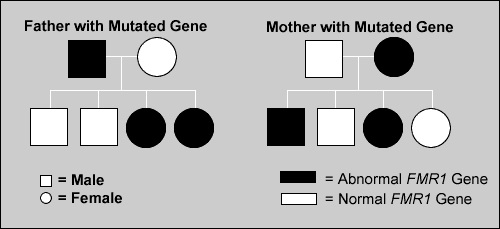
The right side of the diagram depicts the transmission of a fragile x mutation (premutation or full mutation) from a mother to her children. Women have two X chromosomes, and therefore pass one or the other X chromosome to each child. Each child, son or daughter, has a 50% chance of receiving the fragile x mutation a woman carries. The fragile X gene shows anticipation, meaning that with each successive generation, the CGG triplet repeat can increase in number when the gene is passed from the mother. The degree to which a child is affected when inheriting the mutation is determined by whether or not the child is a male or female, and whether or not they inherit the premutation or the full mutation based on the number of repeats inherited.
Researchers have identified that the risk of a premutation between 65-90 CGG repeats expanding to a full mutation (>200 CGG repeats) from mother to child can be personalized by a second test for AGG interruptions within the CGG repeats. The presence of these possible AGG interruptions can stabilize the gene when passed from mother to child, having the greatest impact on the risk of expansion when the mother’s premutation is between 65-90 CGG repeats.