Drug Information
Each of the analytes included in 17 drug groups will be discussed and important features of the drugs will be included. For each analyte the half-life, volume of distribution, pKa, and molecular weight will be listed. Descriptions for what each of these values represent can be found in Table 5. Values were sourced from the Disposition of Toxic Drugs and Chemicals in Man.2
The aspects of each drug groups’ metabolism, abuse, and detection that are of importance will be outlined. Additionally, a guide to the common drug names that contain drug analytes included in the assay can be found in Table 10. All analyte structures were generated using ChemDraw Prime, version 22.2 (©2023 PerkinElmer Informatics, Inc.).
| Elimination Half-Life (t1/2) | Time to reduce concentration of analyte by half |
| Volume of Distribution (Vd) | Theoretical volume where total administered drug in the body equals concentration in plasma (L/kg of body weight) |
| pKa | Negative logarithm of acid dissociation constant; Measure of acidity where lower values represent stronger acids (greater protonation) ([acid] refers to acidic compounds, [base] refers to basic compounds) |
| Molecular Weight (MW) | Mass of chemical compound in grams per mole |
Analgesic drugs have been designed for pain management and possible elimination of pain associated with a number of conditions.3 Drugs considered analgesics can be divided into two subcategories: nonopioids and opioids. Here, the nonopioids or non-steroidal anti-inflammatory or fever reducing drugs (NSAIDs) that are detected in this assay will be outlined. Opioid analgesics will be outlined in a future section. NSAIDs are non-addictive and non-euphoric, as opposed to opiates, which makes them one of the mostly widely used drug classes. The mechanism of action for drugs in this group vary though each have anti-inflammatory or anti-pyretic results.
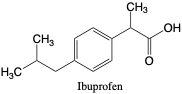 Ibuprofen. Ibuprofen has analgesic, anti-inflammatory, and anti-pyretic properties with highly effective results and few side effects.3 Ibuprofen has been available since 1968 and is sold under various drug names and as an ingredient in other drug products.2 Metabolism of ibuprofen occurs on the isobutyl group. The metabolites 2-carboxyibuprofen and 2-hydroxyibuprofen form after carboxylation or hydroxylation of the carbon side chain, respectively. The parent compound has been found to be completely eliminated in urine after 24 hours.2 Possible side-effects of ibuprofen include nausea, vomiting, blurred vision, gastric pains, diarrhea, and edema.
Ibuprofen. Ibuprofen has analgesic, anti-inflammatory, and anti-pyretic properties with highly effective results and few side effects.3 Ibuprofen has been available since 1968 and is sold under various drug names and as an ingredient in other drug products.2 Metabolism of ibuprofen occurs on the isobutyl group. The metabolites 2-carboxyibuprofen and 2-hydroxyibuprofen form after carboxylation or hydroxylation of the carbon side chain, respectively. The parent compound has been found to be completely eliminated in urine after 24 hours.2 Possible side-effects of ibuprofen include nausea, vomiting, blurred vision, gastric pains, diarrhea, and edema.
- Dose: (oral) 200-400 mg every 4-6 hours
- t1/2: 0.9-2.5 hours
- Vd: 0.14 L/kg
- pKa: 4.4 (acid)
- MW: 206.28 (C13H18O)
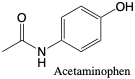 Acetaminophen. Acetaminophen is an anti-fever therapeutic with no anti-inflammatory properties. Acetaminophen metabolizes into a glucuronide and sulfate conjugates and is excreted as about 98% metabolites (2% parent compound).2
Acetaminophen. Acetaminophen is an anti-fever therapeutic with no anti-inflammatory properties. Acetaminophen metabolizes into a glucuronide and sulfate conjugates and is excreted as about 98% metabolites (2% parent compound).2
- Dose: (oral) 350-1000 mg every 4-6 hours
- t1/2: 1-3 hours
- Vd: 0.8-1.0 L/kg
- pKa: 9.5 (acid)
- MW: 151.16 (C8H9NO2)
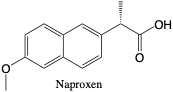 Naproxen. Naproxen has similar qualities as ibuprofen and is commonly administered for the same reasons as ibuprofen or acetaminophen, although it is mainly used for joint pain or pain associated with joint swelling or stiffness. A majority of the dose was found to be cleared 96 hours after taken where 90% was conjugated metabolites and 10% was unchanged.2
Naproxen. Naproxen has similar qualities as ibuprofen and is commonly administered for the same reasons as ibuprofen or acetaminophen, although it is mainly used for joint pain or pain associated with joint swelling or stiffness. A majority of the dose was found to be cleared 96 hours after taken where 90% was conjugated metabolites and 10% was unchanged.2
- Dose: (oral) 250-1500 mg taken 1-3 times daily
- t1/2: 12-36 hours
- Vd: 0.1-0.3 L/kg
- pKa: 5.0 (acid)
- MW: 230.26 (C14H14O)
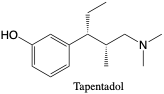 Tapentadol. Tapentadol is a synthetic opioid receptor agonist similar to tramadol. It is metabolized into three major metabolites and excreted in urine over 5 days (3% unchanged).2
Tapentadol. Tapentadol is a synthetic opioid receptor agonist similar to tramadol. It is metabolized into three major metabolites and excreted in urine over 5 days (3% unchanged).2
- Dose: (oral) 50-100 mg every 4-6 hours
- t1/2: 3-7 hours
- Vd: 6-9 L/kg
- pKa: 10.5 (acid)
- MW: 257.80 (C14H24NO)
Tramadol. O-Desmethyl-Tramadol. (Metabolite). Tramadol is a synthetic opioid receptor agonist similar in structure to Tapentadol. The parent compounds and metabolites are excreted in urine over a three-day period, where 29% is unchanged and 20% is O-desmethyl-tramadol.2 O-Desmethyl-Tramadol is detected along with the parent compound by the comprehensive drug assay.
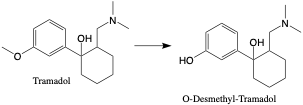
- Dose: (oral) 100-400 mg every 4-6 hours
- t1/2: 4.3-8.3 hours
- O-Desmethyl-Tramadol. t1/2: 6-11 hours
- Vd: 2.6-2.9 L/kg
- pKa: 9.4 (base)
- MW: 263.38 (C16H25NO2)
Local and general anesthetic drugs are relaxants that are used for pain-relief during surgeries. They work by binding to sodium or chloride channels, blocking the passage of impulses that travel through neurons, therefore inhibiting neurological activity. Their ability to block sodium or potassium transport between cells ultimately reduces the electrical impulses through the central nervous system (CNS) that indicate pain.
Abuse
Ketamine was derived from phencyclidine, which has a number of negative toxicological effects and became a popular street drug due to its hallucinogenic properties.2 The use of ketamine has also been found to be abused in many countries over the past decade due to its similarities to phencyclidine.4 Ketamine has been popularized as a “club drug” because of its psychoactive effects, mood controlling properties, and cognitive impairment, but has a number of negative effects including disrupted learning, memory loss, chest pains, and decreased dopamine.4
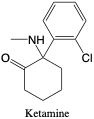 Ketamine. Norketamine (metabolite). Ketamine is a general anesthetic with anti-depressant effects. It is an NMDA receptor antagonist (similar mechanism of action to PCP) that blocks sodium channel activity. It metabolizes to norketamine by N-demethylation and is eliminated over 72 hours as the parent drug, norketamine, and hydroxylated metabolites. Toxic effects of ketamine include hallucinations and general nausea, blurred vision, and vomiting.2
Ketamine. Norketamine (metabolite). Ketamine is a general anesthetic with anti-depressant effects. It is an NMDA receptor antagonist (similar mechanism of action to PCP) that blocks sodium channel activity. It metabolizes to norketamine by N-demethylation and is eliminated over 72 hours as the parent drug, norketamine, and hydroxylated metabolites. Toxic effects of ketamine include hallucinations and general nausea, blurred vision, and vomiting.2
- Dose: (intravenous) 1-4.5 mg/kg,
- (intramuscular) 6.5-13 mg/kg
- t1/2: 2-7 hours
- Vd: 3-5 L/kg
- pKa: 7.5 (base)
- MW: 237.73 (C13H16ClNO)
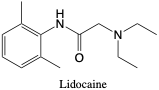 Lidocaine. Lidocaine is a local anesthetic that blocks pain receptors indirectly by binding to sites on the sodium channels in cells, blocking neuronal impulses from transmitting. It is used to reduce pain responses and does not lead to a loss of consciousness. It can be administered as a topical formulation, intravenously, or intramuscularly. It metabolizes into active antiarrhythmic agents that are further metabolized into minor inactive compounds.2 Toxicity of lidocaine manifests as confusion, dizziness, and hypotension and abuse is uncommon.2
Lidocaine. Lidocaine is a local anesthetic that blocks pain receptors indirectly by binding to sites on the sodium channels in cells, blocking neuronal impulses from transmitting. It is used to reduce pain responses and does not lead to a loss of consciousness. It can be administered as a topical formulation, intravenously, or intramuscularly. It metabolizes into active antiarrhythmic agents that are further metabolized into minor inactive compounds.2 Toxicity of lidocaine manifests as confusion, dizziness, and hypotension and abuse is uncommon.2
- Dose: (topical) 2-5% ointment, (intravenous) 50-100 mg, (intramuscular) 300 mg
- t1/2: 0.7-1.8 hours
- Vd: 1.0-1.6 L/kg
- pKa: 7.9 (base)
- MW: 234.34 (C14H22N2O)
Anticonvulsant or antiseizure drugs are used to treat epilepsy and other conditions that could lead to seizures such as bipolar disease. Anticonvulsant and anesthetic drugs have similar mechanisms of action, whereas they prevent neuronal transmission by blockage of sodium or chloride channels. Topiramate, gabapentin, lamotrigine, and pregabalin will be outlined in this section, while benzodiazepines that have similar mechanisms of action will be outlined in a separate section.
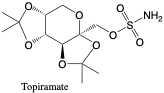 Topiramate. Topiramate is an antiepileptic drug that treats seizures but could also be used to treat alcoholism, psychiatric disorders, migraines, or neuropathic pain.2 It is eliminated in urine over 10 days as the parent compound and six minor metabolites.
Topiramate. Topiramate is an antiepileptic drug that treats seizures but could also be used to treat alcoholism, psychiatric disorders, migraines, or neuropathic pain.2 It is eliminated in urine over 10 days as the parent compound and six minor metabolites.
- Dose: (oral) 100-400 mg taken in 1-2 portions
- t1/2: 30-90 hours
- Vd: 0.5-0.8 L/kg
- pKa: 8.6 (acid)
- MW: 339.36 (C12H21NO8S)
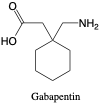 Gabapentin. Gabapentin was synthesized to mimic gamma-aminobutyric acid (GABA), an inhibitory neurotransmitter found in the brain, with structural and mechanistic similarities to pregabalin. It is used to treat epilepsy and works by blocking calcium channels and increasing GABA concentrations in the brain. It is not metabolized and is eliminated renally as the parent compound alone.2
Gabapentin. Gabapentin was synthesized to mimic gamma-aminobutyric acid (GABA), an inhibitory neurotransmitter found in the brain, with structural and mechanistic similarities to pregabalin. It is used to treat epilepsy and works by blocking calcium channels and increasing GABA concentrations in the brain. It is not metabolized and is eliminated renally as the parent compound alone.2
- Dose: (oral) 300-1800 mg taken in 1-3 portions
- t1/2: 5-9 hours
- Vd: 0.8-1.3 L/kg
- pKa: 3.7 (acid)
- MW: 171.24 (C9H17NO2)
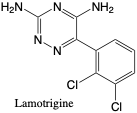 Lamotrigine. Lamotrigine is an antiepileptic drug that is often used in combination with other anticonvulsants. It is metabolized via glucuronidation and eliminated over 6 days, where about 8% remains as the parent compound.2 Adverse reactions to lamotrigine includes skin rash, dizziness, headache, nausea, and vomiting.2
Lamotrigine. Lamotrigine is an antiepileptic drug that is often used in combination with other anticonvulsants. It is metabolized via glucuronidation and eliminated over 6 days, where about 8% remains as the parent compound.2 Adverse reactions to lamotrigine includes skin rash, dizziness, headache, nausea, and vomiting.2
- Dose: (oral) 25 mg taken every other day increased to 100-700 mg in 1-2 portions
- t1/2: 12-62 hours
- Vd: 0.9-1.3 L/kg
- pKa: 5.7 (base)
- MW: 256.09 (C9H7Cl2N5)
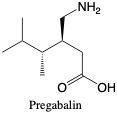 Pregabalin. Pregabalin was synthesized to mimic the neurotransmitter GABA and has structural and mechanistic similarities to gabapentin. It blocks calcium channel activity and increases GABA in the brain, with more activity than gabapentin. It is eliminated over 4 days mainly as the parent drug, and can cause lack of energy, dry mouth, constipation, dizziness, confusion, or blurred vision.2
Pregabalin. Pregabalin was synthesized to mimic the neurotransmitter GABA and has structural and mechanistic similarities to gabapentin. It blocks calcium channel activity and increases GABA in the brain, with more activity than gabapentin. It is eliminated over 4 days mainly as the parent drug, and can cause lack of energy, dry mouth, constipation, dizziness, confusion, or blurred vision.2
- Dose: (oral) 50-200 mg taken thrice daily
- t1/2: 5-11 hours
- Vd: 0.5-0.6 L/kg
- pKa: 4.2 (acid)
- MW: 159.23 (C8H17NO2)
Antidepressant therapeutics work to reduce common side effects for individuals with depression or mood disorders. There is no “cure” for depression, but the drugs included in this category have been known to relieve symptoms such as loss of interest in activities, lower appetite, trouble sleeping, decreased energy, difficulty concentrating, or felling worthless.5 These therapeutics are also commonly used for anxiety disorders, post-traumatic stress disorder (PTSD), and obsessive-compulsive disorder (OCD). There are many subcategories for antidepressants, separated based on the mechanism of action and specific uses for the drugs. Depression, generally, is caused by an imbalance of the neurotransmitters serotonin, dopamine, or norepinephrine in the brain.5
These transmitters travel through synapses between cells to communicate and produce specific responses. Antidepressants block the “reuptake” of neurotransmitters by the presynaptic neuron, allowing for more to bind to postsynaptic receptors in the brain, leading to improved mood. The benefit of the variety of antidepressants on the market and their varying mechanisms of action is that they each target a specific neurotransmitter system. This means if one is not successful for a patient or they are experiencing adverse effects, they can try a drug or combination of drugs with different mechanisms of action. The specific antidepressant analytes and metabolites tested can be found in Table 6.
Selective Serotonin Reuptake Inhibitors (SSRIs)
SSRIs, as implied by their name, block the reuptake of serotonin by neurons, allowing it to build up in the brain and improve an individual’s mood. Drugs that use this mechanism are citalopram, doxepin, fluoxetine, paroxetine, and sertraline. The major active metabolites nordoxepin, norfluoxetine, and norsertraline are detected along with their parent compounds.2 This is currently one of the most popular groups of antidepressants in use today due to their ability to effectively manage depression symptoms with fewer adverse effects.5 Some side-effects that are common include nervousness, headache, sedation, insomnia, or gastrointestinal upset.
Serotonin-norepinephrine Reuptake Inhibitors (SNRIs), Norepinephrine-Dopamine Reuptake Inhibitors (NDRIs), and Serotonin Antagonist and Reuptake Inhibitors (SARIs)
SNRIs, NDRIs, and SARIs block the reuptake of serotonin and norepinephrine, norepinephrine and dopamine, and serotonin, respectively, in the brain. Duloxetine, milnacipran, and venlafaxine are included in the SNRI group and have common adverse effects such as CNS depression, nausea, headache, hypertension, and insomnia.5 The NDRI bupropion has additional side-effects including anorexia and seizures and should not be taken in combination with monoamine oxidase inhibitors (MOAIs). The SARI trazadone has similar side-effects as the other antidepressants and also should not be taken with MAOIs. 1-(3-Chlorophenylpiperazine) is a major metabolite of trazadone with serotonin inhibiting abilities.
Tricyclic Antidepressants (TCAs)
TCAs including amitriptyline, clomipramine, imipramine, maprotiline, and trimipramine were initially synthesized as antihistamines for allergy treatment, but were found to have mood improving results because of their mechanism of action.5 TCAs act on multiple pathways to block serotonin and norepinephrine reuptake, similar to SNRIs, but have more adverse effects and are often only used for severe depression. TCAs are generally not well tolerated and can cause comas or seizures but are currently second-line therapeutic options for multiple disorders.6 Negative effects on cardiac health are common and overdose leading to death has also occurred. The major active metabolites nortriptyline, N-desmethyl-clomipramine, desipramine, and N-desmethyl-trimipramine are detected along with their parent compounds.
Other
The antidepressant aripiprazole that doesn’t fall into any other category can also be detected in urine. Aripiprazole is a dopamine antagonist that can be used as an antidepressant for major depressive disorder or as an antipsychotic.2 It has similar adverse effects compared to other antidepressants including headache, nausea, insomnia, and blurred vision.2
Table 6. Antidepressant analytes and metabolites tested in the comprehensive urine assay and important details considered in their detection.
| Drug Analyte | Metabolites Tested | t1/2 (hours) | Vd (L/kg) |
| SSRIs | |||
| Citalopram | 25-40 | 12-16 | |
| Doxepin | Nordoxepin | 8-25 | 9-33 |
| Fluoxetine | Norfluoxetine | 1-3 days | 20-42 |
| Paroxetine | 7-37 | 3-28 | |
| Sertraline | Norsertraline | 22-36 | 20-50 |
| SNRIs, NDRIs, and SARIs | |||
| Duloxetine | 8-17 | 17-26 | |
| Milnacipran | 7-9 | 3-8 | |
| Venlafaxine | 3-7 | 4-12 | |
| Bupropion | 4-24 | 40 | |
| Trazadone | 1-(3-Chlorphenylpiperazine) | 3-12 | 0.9-1.5 |
| Tricyclic | |||
| Amitriptyline | Notriptyline | 8-51 | 6-10 |
| Clomipramine | N-Desmethyl-Clomipramine | 12-36 | 17 |
| Imipramine | Desipramine | 6-20 | 20-40 |
| Maprotiline | 36-105 | 14-22 | |
| Trimipramine | N-Desmethyl-Trimipramine | 16-39 | 17-48 |
| Aripiprazole | 60-90 | 4.9 |
Antihistamines, by definition, block either histamine release or binding to histamine receptors for treatment of allergy inflammation. Antihistamines that bind to the histamine-1 (H1) receptor in the CNS are divided into first and second generations depending on when they were discovered and their ability to cross the blood-brain-barrier (BBB). First generation antihistamines (ex. diphenhydramine) are more closely related to histamine structurally and have the ability to cross the BBB, causing sedation, drowsiness, and decreased quality of sleep.7
Second generation antihistamines (ex. hydroxyzine) have a decreased ability to cross the BBB, which is beneficial for minimizing sedating effects, and increased selectivity for H1 receptors.7 Second generation antihistamines have antiallergic effects and act on the peripheral instead of the CNS. Antihistamines are inverse agonists for the H1 receptor, and when bound result in the opposite effect than if histamine were bound.7 Antihistamines are considered non-toxic and have very few toxic effects.2
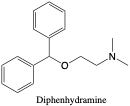 Diphenhydramine. Diphenhydramine, commonly known as Benadryl, is used to relieve allergy symptoms or reactions, and for the common cold, insomnia, vertigo, or motion sickness.
Diphenhydramine. Diphenhydramine, commonly known as Benadryl, is used to relieve allergy symptoms or reactions, and for the common cold, insomnia, vertigo, or motion sickness.
- Dose: (oral) 50-100 mg, (intravenous) 10-50 mg
- t1/2: 3-14 hours
- Vd: 3-4 L/kg
- pKa: 8.3 (base)
- MW: 255.35 (C17H21NO)
 Hydroxyzine. Hydroxyzine is commonly administered to relieve allergic reactions, especially on the skin, or to treat anxiety.
Hydroxyzine. Hydroxyzine is commonly administered to relieve allergic reactions, especially on the skin, or to treat anxiety.
- Dose: up to 400 mg daily
- t1/2: 13-27 hours
- Vd: 13-31 L/kg
- pKa: 2.1 (base), 7.1 (base)
- MW: 374.90 (C21H27ClN2O2)
 Dextromethorphan. Dextromethorphan is a cough medication or antitussive with a different mechanism of action than antihistamines. While it is structurally similar to the opiate morphine, it binds to the N-methyl-D-aspartate (NMDA) receptor, along with others, instead of the opioid receptors the brain.8 It is metabolized to dextrorphan and other minor metabolites within 24 hours.
Dextromethorphan. Dextromethorphan is a cough medication or antitussive with a different mechanism of action than antihistamines. While it is structurally similar to the opiate morphine, it binds to the N-methyl-D-aspartate (NMDA) receptor, along with others, instead of the opioid receptors the brain.8 It is metabolized to dextrorphan and other minor metabolites within 24 hours.
- Dose: (oral) up to 120 mg daily
- t1/2: 3-7 hours
- Vd: 160 L/kg
- pKa: 8.3 (base)
- MW: 271.40 (C18H25NO)
Antipsychotic or neuroleptic drugs can be used for schizophrenia, mania, severe depression, Tourette’s, borderline personality disorder, dementia, and more.9 While there are other treatments for depression, the agents in this category may also be beneficial for those with psychotic behaviors. Antipsychotic agents can be divided into first generation or “typical” neuroleptics and second generation or “atypical” neuroleptics. First generation neuroleptics (ex. chlorpromazine) have calming, mood-stabilizing, and anti-hallucinogenic abilities, and act as antagonists on dopamine receptors in the brain.
Second generation neuroleptics, such as olanzapine and quetiapine are used for both positive side effects (ex. hallucinations) and negative side effects (ex. withdrawal) of mental disorders, and act as dopamine and serotonin antagonists.9 Modafinil, included in this groups, is used to treat narcolepsy. Both first and second generation neuroleptics have weight gain as a major side effect.
 Chlorpromazine. Promazine (Metabolite). Chlorpromazine is one of the most widely used antipsychotic in clinical use today.2 It metabolizes extensively into over 100 metabolites and is excreted slowly.2 One metabolite, promazine, is formed from removal of the chlorine halogen and is detected along with the parent compound.
Chlorpromazine. Promazine (Metabolite). Chlorpromazine is one of the most widely used antipsychotic in clinical use today.2 It metabolizes extensively into over 100 metabolites and is excreted slowly.2 One metabolite, promazine, is formed from removal of the chlorine halogen and is detected along with the parent compound.
- Dose: (oral) 25-100 mg
- t1/2: 7-119 hours
- Vd: 10-35 L/kg
- pKa: 9.3 (base)
- MW: 318.86 (C17H19ClN2S)
 Modafinil
Modafinil
- Dose: (oral) 200-400 mg in 1-2 portions
- t1/2: 9-16 hours
- Vd: 0.85 L/kg
- pKa: unknown
- MW: 273.35 (C15H15NO2S)
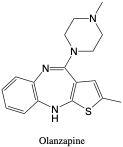 Olanzapine
Olanzapine
- Dose: (oral) 10-20 mg
- t1/2: 21-54 hours
- Vd: 10-20 L/kg
- pKa: 5.0 (base)
- MW: 312.43 (C17H20N4S)
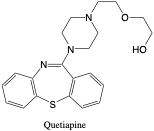 Quetiapine
Quetiapine
- Dose: (oral) 25-400 mg
- t1/2: 2.7-9.3 hours
- Vd: 8-12 L/kg
- pKa: 3.3 (base), 6.8 (base)
- MW: 383.51 (C21H25N3O2S)
Barbiturates are a class of drugs with structural similarities, derived from barbituric acid, that have depressive effects. There are a number of uses for barbiturates including as sedatives, hypnotics, anticonvulsants, or anesthetics, although due to their high toxicity they are not used as widely as benzodiazepines.10 They are primarily used as anesthetics and antiseizure drugs, and often lead to tolerance or dependence due to their mechanism of action on the CNS. Barbiturates have a mechanism of action similar to benzodiazepines, inhibiting the GABA system by binding to chloride channels. Barbiturates can be short or long acting, which significantly changes the number of metabolites and detection window.
Metabolism
Barbiturates are metabolized into inactive substituents with decreased lipophilicity through multiple pathways. Parent compounds are oxidized into alcohols, glucuronidated, de-alkylated at the nitrogen group, or desulfurized.10 Phenobarbital is an intermediate metabolite of mephobarbital that is also administered as a therapeutic. Other major metabolites are not detected in the comprehensive urine assay, but parent compounds may be detected for up to 3 weeks in urine depending on if the drug administered was short or long acting.
Abuse
Barbiturates have a high dependence and abuse potential, and therefore are placed under scheduled status by the Drug Enforcement Administration. Scheduled drugs are regulated to minimize the possibility for abuse and are only available for prescription use. Pentobarbital and secobarbital are Schedule II drugs, butabarbital and butalbital are Schedule III drugs, and phenobarbital is a Schedule IV drug.11
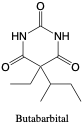 Butabarbital. Butabarbital is an intermediate-acting barbiturate that is prescribed for use as a sedative or hypnotic.2
Butabarbital. Butabarbital is an intermediate-acting barbiturate that is prescribed for use as a sedative or hypnotic.2
- Dose: (oral) 15-30 mg daytime, 50-100 mg nighttime
- t1/2: 34-42 hours
- Vd: unknown
- pKa: 7.9 (acid)
- MW: 234.23 (C10H16N2O3)
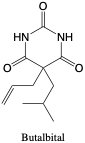 Butalbital. Butalbital is a short-acting barbiturate, currently available for use in combination with other drugs such as acetaminophen, aspirin, caffein, or codeine.2
Butalbital. Butalbital is a short-acting barbiturate, currently available for use in combination with other drugs such as acetaminophen, aspirin, caffein, or codeine.2
- Dose: (oral) 30-100 mg, max 300 mg daily
- t1/2: 35-88 hours
- Vd: 0.8 L/kg
- pKa: 7.6 (acid)
- MW: 224.26 (C11H16N2O3)
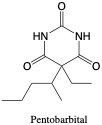 Pentobarbital. Pentobarbital is a short-acting barbiturate with sedative or hypnotic effects, available as a solo drug or in combination with other drugs.2 It is available for oral use as well as intravenous or intramuscular administration.
Pentobarbital. Pentobarbital is a short-acting barbiturate with sedative or hypnotic effects, available as a solo drug or in combination with other drugs.2 It is available for oral use as well as intravenous or intramuscular administration.
- Dose: 15-200 mg
- t1/2: 15-48 hours
- Vd: 0.5-1.0 L/kg
- pKa: 7.9 (acid)
- MW: 226.27 (C11H18N2O3)
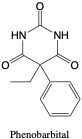 Phenobarbital. Phenobarbital is a long-acting barbiturate often administered as a sedative, anticonvulsant, or for alcohol withdrawl.2 It has been found to lower the blood plasma level of other drugs, and it has limited metabolism where the majority of the drug is eliminated unchanged.2
Phenobarbital. Phenobarbital is a long-acting barbiturate often administered as a sedative, anticonvulsant, or for alcohol withdrawl.2 It has been found to lower the blood plasma level of other drugs, and it has limited metabolism where the majority of the drug is eliminated unchanged.2
- Dose: (oral) 60-200 mg
- t1/2: 2-6 days
- Vd: 0.5-0.6 L/kg
- pKa: 7.2 (acid)
- MW: 232.24 (C12H12N2O3)
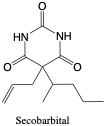 Secobarbital. Secobarbital is a short-acting barbiturate that is administered orally or intravenously as a sedative or hypnotic.2
Secobarbital. Secobarbital is a short-acting barbiturate that is administered orally or intravenously as a sedative or hypnotic.2
- Dose: 8-250 mg
- t1/2: 22-29 hours
- Vd: 1.6-1.9 L/kg
- pKa: 7.9 (acid)
- MW: 238.28 (C12H18N2O3)
Benzodiazepines are a class of drugs commonly used as anxiolytics, sedatives, and hypnotics with similar mechanism of action as barbiturates, and high potential for misuse or abuse.6 Benzodiazepines are often tested in urine drug panels due to their misuse and addiction potential. Depending on acute or chronic use, benzodiazepines may be detected in urine for upwards of 30 days, specifically diazepam, while short acting drugs may only be detecting within 24 hours.6 A complication in detecting benzodiazepines is their extensive metabolism, and being able to detect varying structures is important as there are over 15 commercially available benzodiazepines, and likely more “street drugs”.6 Many assays fail to differentiate structures, leading to false positive results, so being able to detect parent and metabolite compounds is important to confirm certain drugs were taken. While many immunoassays detect benzodiazepines, there is a particularly high potential for false positives or levels falling below cutoffs, and LC-MS/MS is the preferred method of confirmatory analysis. Metabolites detected and important features are outlined in Table 7.
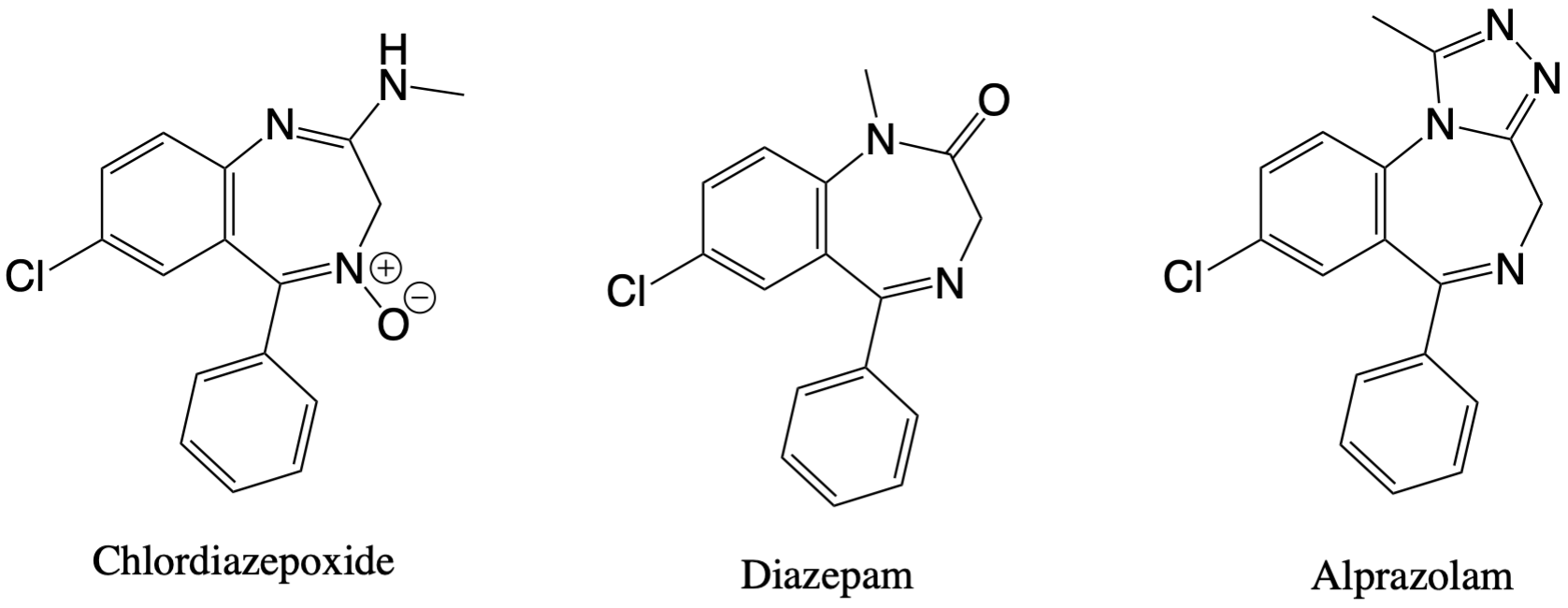
The mechanism of action for all benzodiazepines involve binding to an allosteric site of chloride channels, instead of an active site, modulating GABA binding. The increase of GABA as a result leads to improvements in mood and antianxiety or hypnotic effects. The presence of three rings in benzodiazepine structures results in thousands of variations of substituent groups and different specific binding to the subunits of the GABA receptor complex.10
Metabolism
Benzodiazepines are eliminated as glucuronidated conjugates and have intermediate metabolites that have activity and can be detected (Figure 3).6 The metabolites nordiazepam and temazepam form from either chlordiazepoxide or diazepam, which are further metabolized into oxazepam. Each of these metabolites and the compound lorazepam are metabolized further before excretion. The compounds clonazepam and flunitrazepam are metabolized by an amino group substitution and alprazolam is metabolized into a hydroxylated compound. The metabolites 7-aminoitrazepam and alpha-hydroxytriazolam, formed from nitrazepam and triazolam respectively, are also detected although their parent compounds are not included in the assay. Due to extensive glucuronidation, modified methods involving the addition of beta-glucuronidase to hydrolyze the conjugates can be applied for a more accurate analysis and detection of compounds.6
Abuse
Benzodiazepines are CNS depressants that have potential hypnotic effects and a often leads to high tolerance and dependence. Due to their potential for abuse, benzodiazepines are under Schedule IV by the DEA and are only available for prescription use.11 The most widely used drugs, diazepam and alprazolam, often lead to abuse. The benzodiazepine flunitrazepam is not available for prescription use in the United States and is considered the “date rape” drug, though it can be found in other countries and misused.6
Figure 3. Metabolism of Benzodiazepines.6
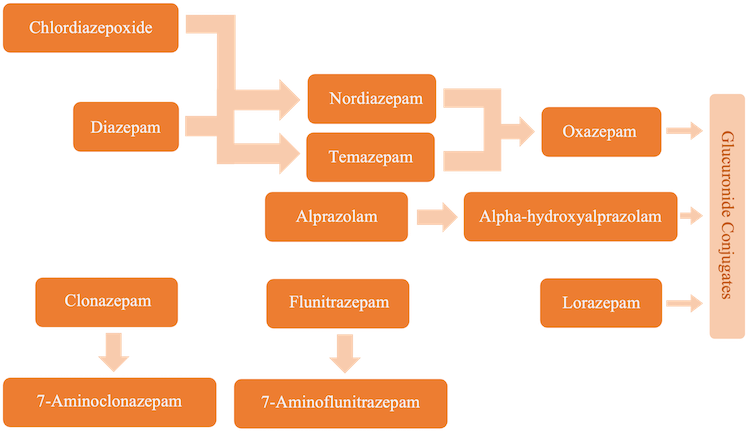
Table 7. Benzodiazepine analytes and metabolites tested in the comprehensive urine assay and important details considered in their detection.
| Drug Analyte | Metabolites Tested | t1/2 (hours) | Vd (L/kg) |
| Alprazolam | Alpha-Hydroxyalprazolam | 6-27 | 0.9-1.3 |
| Chlordiazepoxide | Norchlordiazepoxide | 6-27 | 0.3-0.5 |
| Clonazepam | 7-Aminoclonazepam | 19-60 | 1.5-4.4 |
| Diazepam | Temazepam Nordiazepam Oxazepam | 21-37 | 0.7-2.6 |
| Estazolam | 10-30 | 3.1 | |
| Flunitrazepam | 7-Aminoflunitrazepam | 9-25 | 3.5-5.5 |
| Flurazepam | 2-Hydroxethylfurazepam | 1-3 | 3.4-5.5 |
| Lorazepam | 9-16 | 0.9-1.3 | |
| Midazolam | Alpha-Hydroxymidazolam | 1-4 | 1.0-3.0 |
Cannabinoids are drugs derived from cannabis or marijuana plants that have euphoric effects. While cannabinoids are considered “illicit” drugs, they are one of the most widely used recreational substances.6 Current legislature in the US varies between states from legal recreation use or approved medicinal use only to no legal recreational or medical use. There are currently few medicinally approved uses, though the development of cannabis derived therapeutics is popular in current pharmaceutical research and have shown potential for the future of naturally derived medications.
 Cannabis is ingested in many methods or routes including inhaling smoke or vapors derived from dried flowers or ingesting products that contain marijuana “oils”.6 The main active component of cannabis products is delta-9-tetrahydrocannabinol (THC), though there are 60 other active cannabinoids found in the plants.6 Cannabis products often contain a combination of many components along with delta-9-THC, for example cannabidiol which counters the psychoactive effects of the main active component. While strains of marijuana plants existed at one point in time, current products are most likely from hybrid plants, though they will each have different levels of “indica” or “sativa” that result in different effects when ingested. Due to extensive hybridization of plants, it is hard to characterize the effects of cannabinoids in general, and specific products should be considered unique when considering their activity and side-effects. Cannabinoids’ window for detection varies depending on the frequency of use, ranging from three days to over a month.6 Many synthetic cannabinoids (SC) exist that act on the same cannabinoid receptors and contain active THC, but are not synthesized from marijuana plants. For example, the SC “spice” is dried plant materials that have been sprayed with active cannabinoid components.6 There are many complications when detecting SCs such as their extensive metabolism and varying structures, and therefore they have been removed from the assay.
Cannabis is ingested in many methods or routes including inhaling smoke or vapors derived from dried flowers or ingesting products that contain marijuana “oils”.6 The main active component of cannabis products is delta-9-tetrahydrocannabinol (THC), though there are 60 other active cannabinoids found in the plants.6 Cannabis products often contain a combination of many components along with delta-9-THC, for example cannabidiol which counters the psychoactive effects of the main active component. While strains of marijuana plants existed at one point in time, current products are most likely from hybrid plants, though they will each have different levels of “indica” or “sativa” that result in different effects when ingested. Due to extensive hybridization of plants, it is hard to characterize the effects of cannabinoids in general, and specific products should be considered unique when considering their activity and side-effects. Cannabinoids’ window for detection varies depending on the frequency of use, ranging from three days to over a month.6 Many synthetic cannabinoids (SC) exist that act on the same cannabinoid receptors and contain active THC, but are not synthesized from marijuana plants. For example, the SC “spice” is dried plant materials that have been sprayed with active cannabinoid components.6 There are many complications when detecting SCs such as their extensive metabolism and varying structures, and therefore they have been removed from the assay.
Metabolism
THC is metabolized into hydroxylated components such as 11-OH-THC and THC-COOH, and further into glucuronide conjugates. Very small amounts of the parent compound and the hydroxylated metabolite are excreted in the urine, though after chronic use both are able to be detected.2 The minor metabolites including THC-COOH-glucuronide remain in the body for several weeks before being completely eliminated, even after a single dose.2 The metabolites THC-COOH, 11-OH-THC, and THC-COOH-glucuronide are used as markers for cannabis use in urine.
Although THC is a considered a Schedule I drug federally, and recreation use is not legal in many parts of the country, addiction and abuse is uncommon. Higher doses and prolonged use may exaggerate psychoactive effects and the possibility for developing psychosis.2
THC
- t1/2: 20-37 hours (infrequent use), 3-13 days (frequent use)
- Vd: 4-14 L/kg
- pKa: 10.6 (acid)
- MW: 314.46 (C21H30O2)
Ethanol, also known as ethyl alcohol or alcohol, is a social “drug” that has very little therapeutic use. It is found as the major active component in liquors (20-60% ethanol by volume), fermented beers (3-6% ethanol), wines (10-12%), and more.2 It is also used as an additive to gasoline and in products such as mouthwash in very minimal amounts.
Metabolism
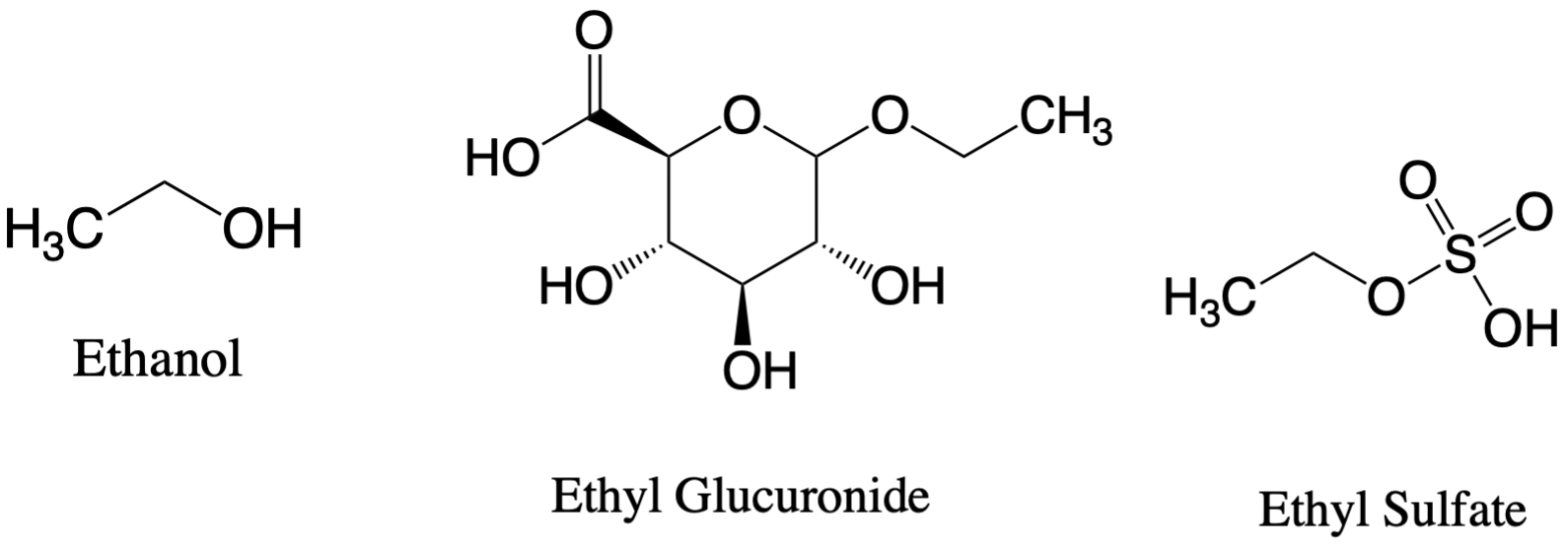
Ethanol is metabolized at a rate independent of the dose consumed, and slightly varying between men and women.2 95% of ethanol consumed is metabolized into acetaldehyde then acetic acid, glucuronide, and sulfate metabolites, usually in similar amounts.2 The ethanol metabolites ethyl glucuronide and ethyl sulfate are detected in urine for 36 hours or longer after ingestion, making them desirable markers for ethanol consumption. After a single dose of ethanol, metabolite concentrations peak at around five or six hours in urine.2 Positive results for both ethyl glucuronide and ethyl sulfate are not required to conclude ethanol was ingested, though the ratio of the two metabolites can indicate whether or not exposure was “incidental” and due to, for example, hand sanitizer or mouthwash.6
Abuse
As alcohol is the most widely consumed substances especially in social settings, there is possibility of addiction and abuse. Acute alcohol use can cause decreased inhibitions and motor coordination, neuropsychic or euphoric effects, and other effects associated with being “drunk”.12 Increased doses of ethanol could lead to a progression of effects as well as memory impairment, instability, or minor coma. Chronic alcohol use can cause lasting damage on the liver, sometimes cause liver disease or cirrhosis.
There is also a higher chance of developing tumors in the organs of the digestive tract, specifically the mouth, larynx, or esophagus, from prolonged and excessive alcohol use. Finally, effects on the heart, brain, and more can develop over time from chronic use. Withdrawal from alcohol after addiction can manifest as hypertension, hallucinations, fever, seizures, and agitation due to the dependence for ethanol developed by the dopamine and GABA neurotransmitter systems. While there are some benefits to ethanol use in moderate amounts, alcohol use or abuse is responsible for millions of deaths every year.12
Illicit drugs are those that have high abuse potential and are Scheduled under either Schedule I or II by the DEA to help prevent their availability. The illicit drugs included in the comprehensive urine drug assay are 3-trifluoromethylphenylpiperazine (TFMPP), 6-monoaceytlmorphine (6-MAM), benzylpiperazine, cocaine, lysergic acid diethylamide (LSD), methamphetamine, methylenedioxymethamphetamine (MDMA), methylenedioxypyrovalerone (MDPV), phencyclidine (PCP), and psilocin. Each have a different structure, mechanism of action, and metabolism that makes their effects and detection unique.
 3-Trifluoromethylphenylpiperazine (TFMPP). TFMPP is a Schedule I drug that has become a popular recreation drug due to its similar effects as MDMA and LSD.2, 11 It is a piperazine derivative with effects on serotonin receptors, leading to euphoric and stimulant effects often persisting for three to six hours.2 Toxic effects may include nausea, vomiting, dizziness, and more after abuse.2
3-Trifluoromethylphenylpiperazine (TFMPP). TFMPP is a Schedule I drug that has become a popular recreation drug due to its similar effects as MDMA and LSD.2, 11 It is a piperazine derivative with effects on serotonin receptors, leading to euphoric and stimulant effects often persisting for three to six hours.2 Toxic effects may include nausea, vomiting, dizziness, and more after abuse.2
- t1/2: 4-8 hours
- Vd: unknown
- pKa: 8.7 (base)
- MW: 230.23 (C11H13F3N2)
6-Monoaceytlmorphine (6-MAM). 6-MAM is the main metabolite of heroin, a Schedule I opioid that is illegal for use in the United States. The street drug is often sold as products containing about 2-6% pure heroin and as cocktails of multiple opioids.2 Heroin (t1/2 = 2-6 min) metabolizes within minutes to 6-MAM (t1/2 = 6-25 min), which is further metabolized to morphine (Figure 4).2 Detection of 6-MAM is likely only possible within 8 hours of use, while morphine is usually detectable within 48 hours of intake.6 For additional information on the mechanism of action of heroin see Opiates.
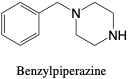 Benzylpiperazine. Benzylpiperazine is a Schedule I drug and piperazine derivative with antidepressant effects.2, 11 It is a common drug of abuse due to its stimulant effects, usually taken in doses of 75-250 miligrams.2 Effects are often felt for six hours and toxicities include increased heart rate or anxiety and nausea.2
Benzylpiperazine. Benzylpiperazine is a Schedule I drug and piperazine derivative with antidepressant effects.2, 11 It is a common drug of abuse due to its stimulant effects, usually taken in doses of 75-250 miligrams.2 Effects are often felt for six hours and toxicities include increased heart rate or anxiety and nausea.2
- t1/2: 4-6 hours
- Vd: unknown
- pKa: 9.3 (base)
- MW: 176.26 (C11H16N2)
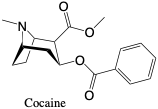 Cocaine. Benzoylecgonine (Metabolite). Cocaine is a Schedule II drug with potent stimulant effects on the central nervous system.2, 11 The active compounded was derived from South American coca leaves and was initially used as a topical anesthetic. Due to its highly active euphoric and stimulant properties, it has become a highly abused drug and is often taken nasally, intravenously, or by smoking doses of 10-120 miligrams.2 The metabolite benzoylecgonine is the main marker for cocaine use, and is detected for up to four days after use.6 The effects of cocaine, including heightened energy and heart rate, can be felt quickly and are addictive. Cocaine use may lead to anorexia, insomnia, or agitation and irritability, depending on doses taken.2, 6 Depression or fatigue can occur as effects ware off. Abuse and overdose are possible, leading to coma or in extreme cases death.
Cocaine. Benzoylecgonine (Metabolite). Cocaine is a Schedule II drug with potent stimulant effects on the central nervous system.2, 11 The active compounded was derived from South American coca leaves and was initially used as a topical anesthetic. Due to its highly active euphoric and stimulant properties, it has become a highly abused drug and is often taken nasally, intravenously, or by smoking doses of 10-120 miligrams.2 The metabolite benzoylecgonine is the main marker for cocaine use, and is detected for up to four days after use.6 The effects of cocaine, including heightened energy and heart rate, can be felt quickly and are addictive. Cocaine use may lead to anorexia, insomnia, or agitation and irritability, depending on doses taken.2, 6 Depression or fatigue can occur as effects ware off. Abuse and overdose are possible, leading to coma or in extreme cases death.
- t1/2: 0.7-1.5 hours
- Vd: 1.6-2.7 L/kg
- pKa: 8.6 (base)
- MW: 303.35 (C17H21NO4)
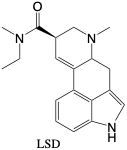 Lysergic Acid Diethylamide (LSD). 2-oxo-3-OH-LSD (Metabolite). LSD is a Schedule I drug with low toxicity that is often abused due to the hallucinations that often result from use.2 11 Only the d-isomer has been found to be a highly active hallucinogen, while the l-isomer has no apparent activity.2 It is often taken orally in doses of only 25-250 micrograms. LSD metabolizes into minor metabolites and 2-oxo-3-OH-LSD, which is detectable for about five days after intake of LSD, though the concentration peaks at 8-16 hours.2 LSD is not addictive and the only major adverse effect is prolonged hallucinations or psychosis.2
Lysergic Acid Diethylamide (LSD). 2-oxo-3-OH-LSD (Metabolite). LSD is a Schedule I drug with low toxicity that is often abused due to the hallucinations that often result from use.2 11 Only the d-isomer has been found to be a highly active hallucinogen, while the l-isomer has no apparent activity.2 It is often taken orally in doses of only 25-250 micrograms. LSD metabolizes into minor metabolites and 2-oxo-3-OH-LSD, which is detectable for about five days after intake of LSD, though the concentration peaks at 8-16 hours.2 LSD is not addictive and the only major adverse effect is prolonged hallucinations or psychosis.2
- t1/2: 2-4 hours
- Vd: 0.28 L/kg
- pKa: 7.8 (base)
- MW: 323.43 (C20H25N3O)
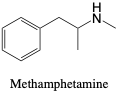 Methamphetamine. Methamphetamine, specifically the d-isomer, is a Schedule II drug of abuse that was once prescribed for its stimulant effects.2 11 The l-isomer is not scheduled and is a main ingredient in inhaled decongestion products, such as Vicks®. Both isomers are detected using the comprehensive urine assay, though the l-isomer from vapor products will likely be present at lower levels when compared to those abusing the d-isomer methamphetamine. Methamphetamine, like other amphetamines, are abused for their euphoric and stimulant effects. Methamphetamine metabolizes into amphetamine and norephedrine, which are also included in the assay for detection, though the parent compounds can be detected in urine for approximately 48 hours.6 Toxic effects of methamphetamine may include dizziness or headache, and more severe adverse effects due to overdose are possible.2 For more information on amphetamine mechanism of action and stimulant effect see Stimulants.
Methamphetamine. Methamphetamine, specifically the d-isomer, is a Schedule II drug of abuse that was once prescribed for its stimulant effects.2 11 The l-isomer is not scheduled and is a main ingredient in inhaled decongestion products, such as Vicks®. Both isomers are detected using the comprehensive urine assay, though the l-isomer from vapor products will likely be present at lower levels when compared to those abusing the d-isomer methamphetamine. Methamphetamine, like other amphetamines, are abused for their euphoric and stimulant effects. Methamphetamine metabolizes into amphetamine and norephedrine, which are also included in the assay for detection, though the parent compounds can be detected in urine for approximately 48 hours.6 Toxic effects of methamphetamine may include dizziness or headache, and more severe adverse effects due to overdose are possible.2 For more information on amphetamine mechanism of action and stimulant effect see Stimulants.
- t1/2: 6-15 hours
- Vd: 3.0-7.0 L/kg
- pKa: 9.9 (base)
- MW: 149.23 (C10H15N)
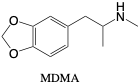 Methylenedioxymethamphetamine (MDMA). MDMA, also known as “Ecstasy” or “Molly” is a derivative of methamphetamine and a Schedule I drug that is often abused for its psychoactive, stimulant effects.2 11 The drug is taken recreationally, and has no approved medical uses. It is often taken orally in doses of 100-150 milligrams.2 MDMA metabolizes into the active metabolite methylenedioxyamphetamine (MDA) and has a peak concentration at around 2 hours after use.2 Toxic effects of MDMA include dizziness, anorexia, headache, anxiety, and insomnia.2
Methylenedioxymethamphetamine (MDMA). MDMA, also known as “Ecstasy” or “Molly” is a derivative of methamphetamine and a Schedule I drug that is often abused for its psychoactive, stimulant effects.2 11 The drug is taken recreationally, and has no approved medical uses. It is often taken orally in doses of 100-150 milligrams.2 MDMA metabolizes into the active metabolite methylenedioxyamphetamine (MDA) and has a peak concentration at around 2 hours after use.2 Toxic effects of MDMA include dizziness, anorexia, headache, anxiety, and insomnia.2
- t1/2: 4-12 hours
- Vd: 3-7 L/kg
- pKa: 8.7 (base)
- MW: 193.24 (C16H21NO3)
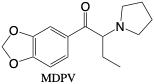 Methylenedioxypyrovalerone (MDPV). MDPV or “bath salts” are a Schedule I drug similar structurally to amphetamines that became a popular drug of abuse for its psychoactivity.2 11 It is ingested as tablets or powders, insufflated nasally, or smoked in doses of 5-20 milligrams, and its effects may last up to 5 hours.2 At high doses, MDPV has similar adverse effects as MDMA.
Methylenedioxypyrovalerone (MDPV). MDPV or “bath salts” are a Schedule I drug similar structurally to amphetamines that became a popular drug of abuse for its psychoactivity.2 11 It is ingested as tablets or powders, insufflated nasally, or smoked in doses of 5-20 milligrams, and its effects may last up to 5 hours.2 At high doses, MDPV has similar adverse effects as MDMA.
- t1/2: 1.3-1.6 hours in rats
- Vd: unknown
- pKa: 8.4 (base)
- MW: 275.34 (C11H13F3N2)
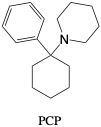 Phencyclidine (PCP). PCP or “angel dust” was initially synthesized based on the structure of ketamine for use as an anesthetic, but became a popular drug of abuse leading to disorientation and hallucinations.2 It is currently a Schedule II drug that can be ingested in a number of routes at doses up to 6 milligrams.2, 11 PCP is able to be detected in urine for up to 8 days as the parent compound.6 The adverse effects caused by PCP use include sedation, hypertension, agitation, seizures, or coma.2
Phencyclidine (PCP). PCP or “angel dust” was initially synthesized based on the structure of ketamine for use as an anesthetic, but became a popular drug of abuse leading to disorientation and hallucinations.2 It is currently a Schedule II drug that can be ingested in a number of routes at doses up to 6 milligrams.2, 11 PCP is able to be detected in urine for up to 8 days as the parent compound.6 The adverse effects caused by PCP use include sedation, hypertension, agitation, seizures, or coma.2
- t1/2: 7-46 hours
- Vd: 5.3-7.5 L/kg
- pKa: 8.5 (base)
- MW: 243.39 (C17H25N)
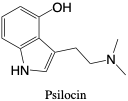 Psilocin. Psilocin is a Schedule I drug of abuse that is the main active metabolite of psilocybin with hallucinogenic effects.2, 11 Both psilocybin and psilocin are derived from a species of mushrooms, where about one gram of mushroom equates to about 10 milligrams of psychoactive ingredients.2 Possible toxic effects of psilocin include anxiety, delusions, disorientation, and hullucinations.2
Psilocin. Psilocin is a Schedule I drug of abuse that is the main active metabolite of psilocybin with hallucinogenic effects.2, 11 Both psilocybin and psilocin are derived from a species of mushrooms, where about one gram of mushroom equates to about 10 milligrams of psychoactive ingredients.2 Possible toxic effects of psilocin include anxiety, delusions, disorientation, and hullucinations.2
- t1/2: 1.8-4.5 hours
- Vd: 2.5-5.0 L/kg
- pKa: 8.5 (base)
- MW: 204.27 (C12H16N2O)
The drugs that don’t seem to fall into any other category are labelled as miscellaneous. These analytes are guanfacine and mitragynine.
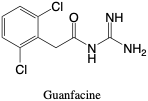 Guanfacine. Guanfacine is an agent used to treat hypertension and ADHD. It is approved for children with ADHD in doses of 1-4 milligrams daily, and adults with high blood pressure in doses of 1-2 milligrams.2 Guanfacine is metabolized by oxidation and further by glucuronidation, though much is excreted unchanged.2 Toxic effects for children may include hallucinations or mania, and in adults include drowsiness, weakness, or headache.2
Guanfacine. Guanfacine is an agent used to treat hypertension and ADHD. It is approved for children with ADHD in doses of 1-4 milligrams daily, and adults with high blood pressure in doses of 1-2 milligrams.2 Guanfacine is metabolized by oxidation and further by glucuronidation, though much is excreted unchanged.2 Toxic effects for children may include hallucinations or mania, and in adults include drowsiness, weakness, or headache.2
- t1/2: 10-30 hours
- Vd: 5-8 L/kg
- pKa: 7.1 (base)
- MW: 246.09 (C9H9Cl2N3O)
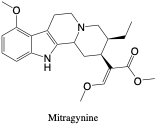 Mitragynine. Mitragynine is a psychoactive component found in the leaves of a native Southeast Asian tree, known as “Kratom”, with analgesic or stimulant effects.2 Leaves are either chewed or dried and powdered for administration of doses up to 15 milligrams taken two or three times daily.2 Mitragynine is eliminated as a variety of metabolites. Possible toxic effects include nausea, fatigue, restlessness, tremors, increased heart rate, and insomnia.2 Dependence may develop from chronic use leading to withdrawal symptoms. Mitragynine has also been administered as an opioid withdrawal treatment as it acts on the opioid receptors.
Mitragynine. Mitragynine is a psychoactive component found in the leaves of a native Southeast Asian tree, known as “Kratom”, with analgesic or stimulant effects.2 Leaves are either chewed or dried and powdered for administration of doses up to 15 milligrams taken two or three times daily.2 Mitragynine is eliminated as a variety of metabolites. Possible toxic effects include nausea, fatigue, restlessness, tremors, increased heart rate, and insomnia.2 Dependence may develop from chronic use leading to withdrawal symptoms. Mitragynine has also been administered as an opioid withdrawal treatment as it acts on the opioid receptors.
- t1/2: 17-29 hours
- Vd: 30-50 L/kg
- pKa: 7.3 (base)
- MW: 398.50 (C23H30N2O4)
Drugs included in the muscle relaxant group are commonly used for a range of moderate to severe musculoskeletal conditions. They can be divided into antispastic treatments for cerebral palsy and multiple sclerosis, or antispasmodic treatments used for back and neck pain, arthritis, or fibromyalgia.13 Baclofen is an antispastic agent while carisoprodol and cyclobenzaprine are antispasmodic agents.
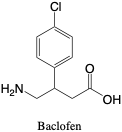 Baclofen. Baclofen is a muscle relaxant used as a therapeutic to alleviate spasticity from multiple sclerosis or cerebral palsy, but primarily as a treatment for alcoholism.2 Baclofen is an agonist for GABA receptors with potential for addiction or abuse. Toxic effects include nausea, sedation, dry mouth, dizziness, headache, and more.2 Chronic users who are withdrawn from use may experience agitation, hallucination, psychosis, fever, or seizures.2 It is eliminated over three days in the urine and the parent compound can be detected within this window.
Baclofen. Baclofen is a muscle relaxant used as a therapeutic to alleviate spasticity from multiple sclerosis or cerebral palsy, but primarily as a treatment for alcoholism.2 Baclofen is an agonist for GABA receptors with potential for addiction or abuse. Toxic effects include nausea, sedation, dry mouth, dizziness, headache, and more.2 Chronic users who are withdrawn from use may experience agitation, hallucination, psychosis, fever, or seizures.2 It is eliminated over three days in the urine and the parent compound can be detected within this window.
- Dose: (oral) 5 mg, 3 times daily for 3 days
- t1/2: 2-8 hours
- Vd: 2-3 L/kg
- pKa: 3.9 (acid), 9.6 (base)
- MW: 213.66 (C10H12ClNO2)
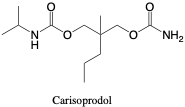 Carisoprodol. Meprobamate (Metabolite). Carisoprodol is a Schedule IV drug that is often used for skeletal muscle conditions and as a sedative.2, 11 It is able to provide relief when supplied alone or in combination with other drugs such as acetaminophen, aspirin, caffeine, or codeine.2 Carisoprodol is metabolized into meprobamate, and both are detected in urine. Carisoprodol is scheduled due to its potential for addiction from chronic use. Toxic effects may include drowsiness, headache, or loss of balance and coordination.2
Carisoprodol. Meprobamate (Metabolite). Carisoprodol is a Schedule IV drug that is often used for skeletal muscle conditions and as a sedative.2, 11 It is able to provide relief when supplied alone or in combination with other drugs such as acetaminophen, aspirin, caffeine, or codeine.2 Carisoprodol is metabolized into meprobamate, and both are detected in urine. Carisoprodol is scheduled due to its potential for addiction from chronic use. Toxic effects may include drowsiness, headache, or loss of balance and coordination.2
- Dose: (oral) 350 mg, up to 4 times daily
- t1/2: 0.9-2.4 hours
- Vd: 0.9-1.3 L/kg
- pKa: 4.2 (acid)
- MW: 260.33 (C12H24N2O4)
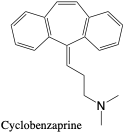 Cyclobenzaprine. N-Desmethyl-Cyclobenzaprine (Metabolite). Cyclobenzaprine is a centrally-acting skeletal muscle relaxant that is structurally similar to amitriptyline (see Antidepressants).2 One of the many metabolites of Cyclobenzaprine is N-desmethyl-cyclobenzaprine which is detected in urine along with the parent compound over a 5 day period. Toxic effects include drowsiness, dry mouth, dizziness, blurred vision, nausea, and more.2 Abuse and overdose may occur, causing hallucinations, coma, and cardia arrhythmia.2
Cyclobenzaprine. N-Desmethyl-Cyclobenzaprine (Metabolite). Cyclobenzaprine is a centrally-acting skeletal muscle relaxant that is structurally similar to amitriptyline (see Antidepressants).2 One of the many metabolites of Cyclobenzaprine is N-desmethyl-cyclobenzaprine which is detected in urine along with the parent compound over a 5 day period. Toxic effects include drowsiness, dry mouth, dizziness, blurred vision, nausea, and more.2 Abuse and overdose may occur, causing hallucinations, coma, and cardia arrhythmia.2
- Dose: 10-20 mg, up to 3 times daily
- t1/2: 20-40 hours
- Vd: unknown
- pKa: 8.5 (base)
- MW: 275.39 (C20H21N)
Nicotine is a highly addictive component derived from tobacco plants, now used as an ingredient in cigarettes, cigars, and other tobacco products where 0.5-8.0% is nicotine by weight.2 Use results in stimulation of the central nervous system and adverse effects including nausea, vomiting, sweating, salivation, and dizziness.2 At lethal doses from 30 to 60 milligrams, convulsion, paralysis, or death may occur.2
When smoking nicotine containing products into the air of public spaces causes second-hand exposures that often results in nicotine inhalation. Detection of second-hand inhaled nicotine in urine is possible, although the metabolite tested will often fall below the LLOQ of 100 ng/mL. Electronic or “e-cigarettes” have become increasingly popular and contain nicotine solutions of 4-36 mg/mL that is heated by a battery for inhalation.2
Metabolism
The first step of the metabolism of nicotine is oxidation into cotinine, which is further metabolized into oxidated, dealkylated, and ring-cleaved compounds. The initial metabolite cotinine is detected in urine to confirm nicotine use.
Abuse
Nicotine is highly addictive due to its effects on the dopamine system in the brain and overuse causing a number of side-effects including cancer is possible. Products designed as therapy for those addicted to nicotine contain smaller amounts of the active ingredient in lozenge, tablet, gum, nasal spray, inhaler, or patch formulations. These products are able to minimize the effects of withdrawal and eventually could lessen dependence on nicotine products.
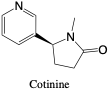 Cotinine
Cotinine
- t1/2: 19 hours
- Vd: (nicotine, intravenous) 1.0-3.0 L/kg
- pKa: 4.8 (base)
- MW: 176.22 (C10H12N2O)
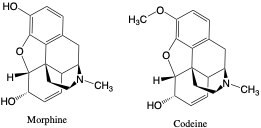 Drugs belonging in the opioid group act on the opioid receptor of the central nervous system, resulting in analgesic effects for pain management. The term opiate refers to morphine and codeine which are naturally derived from the poppy plant.14 “Opioids” are synthetic opiates that bind to opioid receptors but are not naturally derived. These include heroin, fentanyl, hydromorphone, oxymorphone, hydrocode, and more. Opioids are used for moderate to severe pain but have a number of adverse effects and high abuse potential.2
Drugs belonging in the opioid group act on the opioid receptor of the central nervous system, resulting in analgesic effects for pain management. The term opiate refers to morphine and codeine which are naturally derived from the poppy plant.14 “Opioids” are synthetic opiates that bind to opioid receptors but are not naturally derived. These include heroin, fentanyl, hydromorphone, oxymorphone, hydrocode, and more. Opioids are used for moderate to severe pain but have a number of adverse effects and high abuse potential.2
Immunoassays are generally not successful at separating opioids, as there are many with similar structures, so LC-MS/MS analysis is required to fully understand which of the opioids may have been used. The detection of opioids and metabolites is complicated when considering street drugs that often contain a combination of multiple analytes. False positive codeine or morphine results are also possible if a large amount of poppy seeds are consumed, especially if they are unwashed.
Substances that have high abuse potential are placed into “scheduled” groups by the DEA depending on their illicit and medical uses. The opiates morphine and codeine are scheduled but are available for prescription use depending on their concentrations. Heroin is a “Schedule I” drug which means it is not deemed medically useful and is not provided as a therapeutic in the United States.6 Heroin is difficult to detect using LC/MS, although it is metabolized to 6-MAM and morphine which can be detected. This is outlined further under Illicit drugs. The opioids detected, their metabolites, schedule status, and other important values are listed in Table 8.
Oxycodone
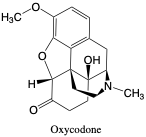 Oxycodone is a semisynthetic opioid that metabolizes into the active metabolite oxymorphone and the inactive metabolite noroxycodone.6 These metabolites can also be detected by the comprehensive drug assay most accurately within 4 days of ingestion.6 This metabolism is separate from the opiates codeine and morphine (Figure 4). It has a high abuse potential, like other opioids, and is a Schedule II drug that is only available for prescription use. Oxycodone has comparable potency to morphine, although the efficacy (pain-relieving abilities) is greater.2 It is often given in combination with acetaminophen, ibuprofen, naloxone, caffeine, or other components for oral administration.2 Oxycodone has similar toxic effects to other opioids including nausea, vomiting, dizziness, diaphoresis, and more.2
Oxycodone is a semisynthetic opioid that metabolizes into the active metabolite oxymorphone and the inactive metabolite noroxycodone.6 These metabolites can also be detected by the comprehensive drug assay most accurately within 4 days of ingestion.6 This metabolism is separate from the opiates codeine and morphine (Figure 4). It has a high abuse potential, like other opioids, and is a Schedule II drug that is only available for prescription use. Oxycodone has comparable potency to morphine, although the efficacy (pain-relieving abilities) is greater.2 It is often given in combination with acetaminophen, ibuprofen, naloxone, caffeine, or other components for oral administration.2 Oxycodone has similar toxic effects to other opioids including nausea, vomiting, dizziness, diaphoresis, and more.2
Metabolism
The metabolism of opioids is quite complicated and, when understood, can benefit clinicians in their knowledge of what substances were taken by patients (Figure 4). For example, the presence of codeine and morphine in urine likely means codeine was used, but morphine alone could mean codeine, heroin, or morphine were taken. Testing for opioids in urine often requires the presence of metabolites to ensure it is not a false positive result, and often only the metabolite can be detected and conclusions about the initial substance taken cannot be made. The metabolism of opioids including the metabolites tested in this assay are outlined in Figure 4.
Codeine is metabolized into morphine and along with other minor and glucuronide metabolites, including norcodeine and hydrocodone. Morphine is metabolized into glucuronide conjugates as well as hydromorphone and can be detected in urine for up to 4 days.14 Heroin is metabolized quickly into 6-MAM (t1/2 = 36 minutes)6 and then into morphine, usually within 24 hours.14 Hydromorphone is a semisynthetic opioid, along with being a minor metabolite of morphine, codeine, and hydrocodone, that can be detected for up to 4 days. The synthetic opioid methadone metabolizes into inactive EDDP, and both are often detected in urine assays.6 Methadone has a long half-life (15-55 hours) and can be detected for longer than the other opioids. Other synthetic opioids such as fentanyl, meperidine, naltrexone, and propoxyphene have major metabolites that are also detected by LC-MS/MS.
Abuse
Opioids are commonly abused due to their addictive nature and euphoric effects. Abusers often develop a tolerance, requiring increased doses to feel the effects and ultimately leading to addiction and abuse.14 All opioids have the possibility for abuse, which is why many have a scheduled status (Table 8). Scheduled drugs are regulated due to their high dependance potential. Those under Schedule I have the highest abuse potential are considered to have no medical use, such as the illicit drugs cocaine and heroin. Toxic effects of opioid use or overuse include nausea and vomiting, sedation, and dizziness.2
Figure 4. Metabolism of opioids:
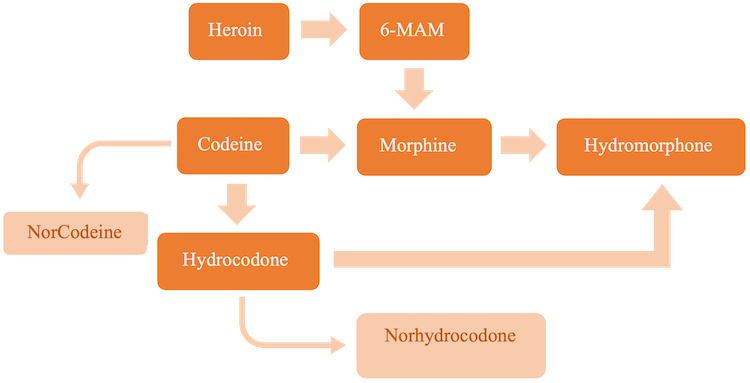
Table 8. Opioids and metabolites tested in the comprehensive urine assay and important details considered in their detection
| Drug Analyte | Metabolites Tested | Schedule Status11 | t1/2 (hours) | Vd (L/kg) |
| From Opium | ||||
| Codeine | NorCodeine | Schedule II | 1.2-3.9 | 2.5-3.5 |
| Morphine | Schedule II | 1.3-6.7 | 2-5 | |
| Semisynthetic | ||||
| Buprenorphine | Norbuprenorphine | Schedule III | 2-4 | 1.4-6.2 |
| Hydrocodone | Norhydrocodone Hydromorphone Dihydrocodeine | Schedule II | 3.4-8.8 (Hydromorphone, 3-9) | 3.3-4.7 |
| Heroin | 6-MAM | Schedule I | 2-6 minutes | 25 |
| Oxycodone | Oxymorphone Noroxycodone | Schedule II | 3-6 | 1.8-3.7 |
| Synthetic | ||||
| Alfentanil | Schedule II | 1-2 | 0.3-1.0 | |
| Butorphanol Tartrate | Schedule IV | 2.9-8.4 | 5-10 | |
| Fentanyl | Despropionylfentanyl Norfentanyl | Schedule II | 3-30 | 3-8 |
| Meperidine | Normeperidine | Schedule II | 2-5 | 3.7-4.2 |
| Methadone | EDDP | Schedule II | 15-55 | 4-7 |
| Nalbuphine | Schedule IV | 1.9-7.7 | 2.4-7.3 | |
| Naloxone | Schedule VI | 0.5-2.5 | 2.6-2.8 | |
| Naltrexone | Naltrexol | Unscheduled | 1-3 (IV) 8-10 (oral) | 14-24 |
| Pentazocine | Schedule IV | 2.1-3.5 | 4.4-7.8 | |
| Propoxyphene | Norpropoxyphene | Schedule IV | 8-24 | 12-26 |
| Sufentanil | Schedule II | 1.6-6.3 | 1.5-3.9 | |
Stimulant drugs are those with stimulant effects on the central nervous system, including amphetamine, mephedrone, methylphenidate, phentermine, and pseudoephedrine. These drugs are used for medical reasons, such as for ADHD, obesity, asthma, narcolepsy, hypotension, or congestion although they are often used recreationally or for performance enhancement, specifically by athletes.15 The stimulant amphetamine has a number of non-therapeutic derivatives that are not used as medical therapeutics such as MDMA (illicit), MDPV (illicit), and mephedrone.
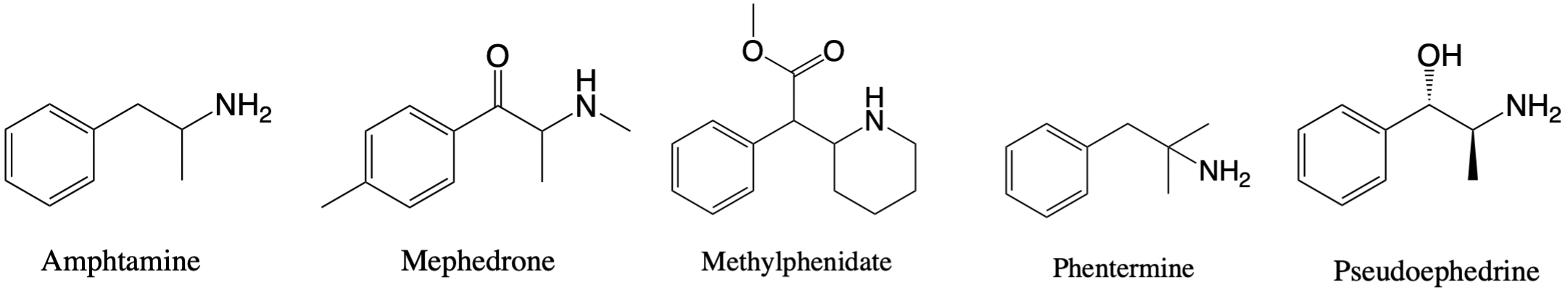
The mechanism of action for stimulants involves an increase in the catecholamines norepinephrine and dopamine. The effects of this mechanism include increased energy, mental stimulation, euphoria, and higher cognition.15 Toxic effects of stimulant use may include decreased appetite, anxiety, headaches, weight loss, insomnia, sweating, seizures, chest pain, and more.2, 15 The analytes included in the stimulant category, their metabolites, schedule status, and other important values can be found in Table 9.
Metabolism
The metabolism of amphetamine includes the minor metabolite norephedrine, which is bioactive and contributes to onset of effects especially from chronic use.2 Both amphetamine and norephedrine may be detected in urine for several days after use.2 Methylphenidate is metabolized rapidly into ritalinic acid, an inactive metabolite that is detected for longer than the parent compound. Phentermine and pseudoephedrine are mainly eliminated unchanged in urine.
Abuse
Due to the euphoric effects and potential for abuse, many stimulants are under Scheduled status by the DEA, and are only available for prescription use (Table 9). Amphetamines have also been abused by athletes for its performance enhancing abilities. Toxic effects from chronic stimulant use can be treated using benzodiazepines, beta-blockers, or other antihypertensive medications.15
Table 9. Stimulant analytes and metabolites tested in the comprehensive urine assay and important details considered in their detection.
| Drug Analyte | Metabolites Tested | Schedule Status11 | t1/2 (hours) | Vd (L/kg) |
| Amphetamine | Norephedrine | Schedule II | 7-34 | 3.2-5.6 |
| Mephedrone | Schedule I | 1.8-2.6 | Unknown | |
| Methylphenidate | Ritalinic acid | Schedule II | 1.4-6.2 | 11-33 |
| Phentermine | Schedule IV | 19-24 | 3-4 | |
| Pseudoephedrine | Unscheduled | 3-16 | 2.0-3.0 |
Z-drugs
Z-drugs are hypnotic agents that are often used therapeutically to treat insomnia. Z-drugs include zaleplon, zolpidem, and zopiclone. These drugs were developed in order to improve adverse effects from benzodiazepine use. They act on the same GABA receptors of benzodiazepines and are central nervous system depressants.16 They have a rapid onset, usually within 30 minutes, and toxic effects including hallucinations, amnesia, parasomnia, headache, gastrointestinal upset, and dizziness.16 Abuse and overdose are less likely from z-drugs than benzodiazepines, though withdrawal symptoms are similar. Chronic use may lead to anxiety, tremors, palpitations, seizures, and in severe cases death. Z-drugs are able to be detected in urine for up to 48 hours, or 72 hours from higher doses, as the parent compound.
 Zaleplon
Zaleplon
- Dose: (oral) 5-20 mg
- t1/2: 0.9-1.2 hours
- Vd: 1.0-1.5 L/kg
- pKa: 0.3 (base)
- MW: 305.33 (C17H15N5O)
 Zolpidem
Zolpidem
- Dose: (oral) 5-12.5 mg
- t1/2: 1.4-4.5 hours
- Vd: 0.5-0.7 L/kg
- pKa: 6.2 (base)
- MW: 307.39 (C19H21N3O)
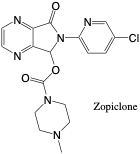 Zopiclone
Zopiclone
- Dose: (oral) 3.75-15 mg
- t1/2: 3.5-6.5 hours
- Vd: 1.3-3.6 L/kg
- pKa: 6.7 (base)
- MW: 388.81 (C17H17ClN6O3)
Table 10. Analytes contained in common drugs, and their trade names.
| Chemical Name | Common/Trade Name |
| Analgesic | |
| Ibuprofen | Advil® |
| Acetaminophen | Tylenol® |
| Naproxen | Aleve® |
| Tapentadol | Nucynta®, |
| Tramadol | ConZip®, Ultram® |
| Anticonvulsant | |
| Topiramate | Qudexy®, Topamax® |
| Gabapentin | Gralise®, Horizant® |
| Lamotrigine | Lamictal® |
| Pregabalin | Lyrica® |
| Antidepressants | |
| Carisoprodol | Soma® |
| Bupropion | Wellbutrin® |
| Doxepin | Silenor®, Zonalon® |
| Duloxetine | Cymbalta® |
| Fluoxetine | Prozac®, Sarafem® |
| Paroxetine | Paxil® |
| Sertraline | Zoloft ® |
| Antihistamines | |
| Dextromethorphan | NyQuil®, Robitussin® |
| Diphenhydramine | Benadryl® |
| Hydroxyzine | Vistaril® |
| Barbiturates | |
| Butabarbital | Butisol® |
| Butalbital | Fioricet®, Fiorinal® |
| Pentobarbital | Nembutal® |
| Secobarbital | Seconal® |
| Phenobarbital | Luminal®, Solfoton® |
| Benzodiazepines | |
| Alprazolam | Xanax® |
| Clonazepam | Klonopin® |
| Diazepam | Valium® |
| Lorazepam | Ativan® |
| Midazolam | Versed® |
| Guanfacine | Intuniv® |
| Muscle Relaxants | |
| Baclofen | Lioresal® |
| Carisoprodol | Soma® |
| Cyclobenzaprine | Flexeril® |
| Opiates | |
| Hydromorphone | Dilaudid® |
| Methadone | Dolophine® |
| Meperidine | Demerol® |
| Oxycodone | OxyContin®, Percocet® |
| Fentanyl | Sublimaze®, Duragesic® |
| Stimulants | |
| Amphetamine | Adderall®, Dexedrine® |
| Methylphenidate | Ritalin® |
| Phentermine | Adipex®, Lomaira® |
| Pseudoephedrine | Sudafed® |
| Z-Drugs | |
| Zaleplon | Sonata® |
| Zolpidem | Ambien® |
| Zopiclone | Imovane® |
Anesthesiology (SOM)
CU Anschutz
Leprino Building
12401 East 17th Avenue
7th Floor
Aurora, CO 80045
iC42 Testing Locations
Partners
CMS Login