lyons research lab
Postpartum Breast Cancer
Research at the University of Colorado Anschutz Cancer Center focuses on understanding how breast cancer cells survive, adapt, and metastasize, with particular attention to postpartum breast cancer, an aggressive subtype diagnosed after childbirth. During the postpartum period, the mammary gland undergoes involution, a normal remodeling process that generates a wound like, inflammatory microenvironment. Studies have shown that breast tumor cells can exploit this temporary physiological state, leveraging involution associated cues to enhance survival, invasion, and resistance to therapy. These findings are especially important given the significantly increased risk of metastasis and poorer clinical outcomes observed in postpartum breast cancer.
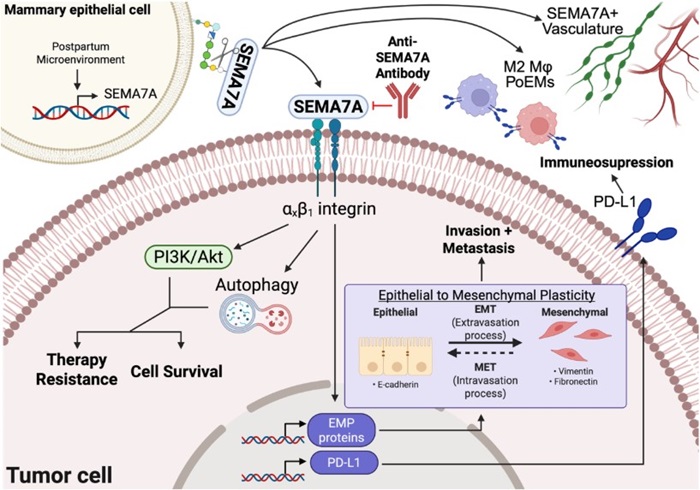
Key Research Focus: Semaphorin 7A (SEMA7A)
A central area of investigation is the role of Semaphorin 7A (SEMA7A) as a major regulator of tumor cell adaptation during postpartum involution. Research has demonstrated that SEMA7A functions as a broad mediator of breast cancer progression, supporting tumor cells through multiple mechanisms, including:
- Resistance to cell death.
- Activation of pro survival and cellular stress response pathways, including autophagy.
- Recruitment and activation of macrophages.
- Promotion of lymphangiogenesis.
- Enhancement of immunosuppression.
- Suppression of tumor inhibitory programs.
These mechanisms enable cancer cells to survive in hostile environments-such as the bloodstream or distant tissues-ultimately facilitating metastatic spread. SEMA7A has also been identified as both a biomarker of recurrence and a promising therapeutic target in postpartum breast cancer and in all breast cancers where it is expressed.
In partnership with Pearl Scientific, Inc., a novel antagonistic SEMA7A antibody has been developed and is currently undergoing humanization with the goal of enabling future clinical trials in ER+ and ER- breast cancers.
Approach
This research program integrates molecular, cellular, and in vivo experimental systems to define how cancer cells co-opt normal physiological processes-particularly those associated with postpartum mammary gland involution-to promote disease progression. By uncovering the adaptive pathways that allow tumor cells to survive and metastasize, the work aims to identify critical vulnerabilities that can be leveraged to develop new therapeutic strategies. This approach ultimately supports the broader goal of improving outcomes for individuals with postpartum breast cancer as well as those with breast cancers that express SEMA7A.
lab personnel
Traci Lyons, PhD
Principle Investigator
Alexandria Becks, BS
PhD Candidate
Lauren Cozzens, BS
PhD Candidate
Heather Fairchild, MS
Senior Research Services Professional
Veronica Wessells, BA
Research Services Professional